[1] [1]
Tübingen, September 1999 1
1.1 Clover in day- and night-position
1.2 Fragrence of Exacum affine as function of day time
1.3 Golden hamsters at night and at day
1.4 The drinking duck
1.5 Desmodium rhythm: illustration
1.6 Desmodium-rhythm: curve
1.7 Desmodium-rhythm: recording
1.8 Desmodium-rhythmicity: temperature effect
1.9 Example from a laboratory book: content
1.10 Example from a laboratory book: entrances
1.11 Example from `Science Citation Index'
1.12 Chronobiological journals
1.13 Current Contents
2.1 Imaging with video camera, framegrabber and computer
3.1 Parameters of an oscillation
3.3 Stomatal movement rhythm of Vicia
3.2 1012 plus2 minus5 @ plus3 plus3 minus3 plus2 minus plus3 minus5 plus2 minus plus2 minus2 plus2 minus Phase diagram of Desmodium
3.5 Period determination with a fitting line
3.4 Frequency folding
3.6 Signal average method
4.1 Predator-prey model
4.2 Feedback model
4.3 Phase diagram of oscillations of a predator-prey system
5.1 Chemical oscillator
5.2 Belousov-Zhabotinsky reaction scheme
5.6 Stomatal apparatus
5.8 Oscillations of transpiration in oat leaf: Feedback loop
5.9 Oat seedling with primary leaf
5.3 Wave pattern of Belousov-Zhabotinsky reaction
5.4 Gravitropic pendulum
5.5 Gravitropic pendulum: curve
5.7 Rhythmic transpiration in oat
6.1 Kalanchoe-flowers
6.2 pH rhythm in CAM plants
6.4 Recording of Oxalis leaf movement
6.3 Oxalis cuvette
6.5 Anatomy of Oxalis pulvinus
6.6 Anatomy of Kalanchoe flower
6.7 Petal movement of Kalanchoe and osmotic pressure
6.8 Recording of Kalanchoe petal movement
6.9 Kalanchoe petal movement: curve
7.1 Synchronized Thalassomyxa culture
7.2 Thalassomyxa rhythmicity
8.1 Examples for day- and night active animals
8.2 Actogram of a housefly
8.3 Housefly
8.4 Recording system
8.5 fly cage
8.6 Recording the activity of Drosophila
8.8 Recording system for eclosion rhythm of Drosophila
8.7 Special forceps
8.9 Soot method for eclosion rhythm of Drosophila
8.10 Soot method for eclosion rhythm of Drosophila: Example of curves
8.12 Temperature course and sleep-wake periods of a person
8.11 Sleep-wake cycle of a baby
8.13 `Average day' of rectal temperature of a person
8.14 Circadian rhythm and chronobiological phase type
9.2 Critical darkperiod of Pharbitis
9.1 Flower buds and vegetative buds of Pharbitis nil
9.3 Exhaustor
1.2 Time of maxima, minima and points of deflection of the Desmo -dium lateral leaflet movement
1.3 Time and standard deviation (SD) of maxima, minima and points of inflection (PI) of the Desmo -dium lateral leaflet movement.
1.4 Table for recorded values of Desmodium motorium lateral leaflet movement.
8.1 Evaluation of the chronobiological phase type from the results of the netherlandic questionair.
8.2 Chronobiological phase type of different persons. xx
Many processes in organisms are rhythmic. Rhythms with periods of about 24 hours are found already in the lowest organisms such as cyanobacteria. In unicellulars, fungi, plants and animals including humans they are widespread. Our knowledge of such rhythmic processes is increasing constantly by new observations and experimental results. But the underlying mechanisms are not yet clarified in a single case and are therefore a challenge for scientists.
This new area of `Chronobiology' is not only of interest to scientists. Even laymen, pupils and students find it intriguing. Furthermore, these rhythms in organisms can be observed and experiments performed by them, because simple procedures are often sufficient.
In the natural sciences it is important to learn scientific methods and to practice them. We believe, that this fascinating research area of chronobiology is useful for it. In order to get familiar with the field and the methods used in it we have published a few years ago a workbook [39]. It was written to facilitate the observation of and experimenting with rhythms in biological systems. For many of the proposed experiments special equipment was necessary. This can nowadays be replaced by computers. They are much more versatile and also usable for analysing the data. Often they are cheaper than the special equipment used before. We have therefore written this book. It was used as an instruction for biology students of the university of Tübingen attending courses in chronobiology. It might, however, be of interest also to students of other universities and to teachers at high schools, pupils and other people.
We hope that this book rises interest for this fascinating new area of life sciences. It should help to study rhythms more closely. If it furthermore mediates a little bit of the excitement and enthusiasm which results from solving problem s in science, its aim is fullfilled. Another book will be published here soon which serves as an introduction into the field of chronobiology.
To satisfy the curiosity of young people is unfortunately nowadays not much promoted, because schools stick often too tightly to the curiculum and offer too much matter of teaching. This leads easily to only secondarily motivated students for whom the scores are the main aim of going to school. It was the great pedagogue Pestalozzi who once stated:
All unsere Erziehung ist nicht einen Heller wert,
wenn Mut und Freude dabei verloren gingen2
Alle Schöpfung schwingt im Reigen,
Freude heisst ihr hohes Lied.
Nur der Mensch will sich nicht beugen,
jagt nach fremdem Glück sich müd.
Freunde, sucht den Sinn der Dinge,
dass auch Freude euch durchdringeJ.W. v. Goethe
All complicated systems tend to oscillate. Machines, bridges, electronic equipment are examples. The time it takes for one cycle (`period lengths'3) is the result of the properties of the system. It usually does not correspond to the period length of a rhythmic event in the physical environment.
Organisms as another example of complicated systems are also prone to oscillate. This ability is found at all levels of organisation. Plants and animals have adapted themself during the course of evolution to the time structure of their environment. This is periodic: The 24 hour day-night rhythm (daily rhythms), the 24.8, 12.4 and monthly rhythm (tidal and lunar rhythms) brought about by th orbit of the moon, or 12 months rhythms (annual rhythms, photoperiodism) caused by the orbit of the earth around the sun. Corresponding rhythms are found in numerous organisms.
This book consists of three parts. In the first part we describe methods and equipment. They are the basis for planning, execution and analysis of experiments. Many of the proposed experiments can be done by using a computer in connection with a video camera and a frame grabber (also called digitizer)4. In this way the experimental data can be recorded and analyzed. The system and the programs are mentioned briefly . They are described more detailed in a handbook [36] and in a book [37]. A system to record the locomotor activity of animals with infrared light beams is also described. In a special chapter we mention the importance of modelling, which allows the simulation of biological rhythms. We will also get a glimpse of the different methods to analyze rhythms (`time series analysis').
In the second part experiments are proposed. Some short term oscillations which are not adjustments to external periodicities (`ultradian rhythms') will be described: a chemical oscillator, the gravitropic pendulum, a transpiration rhythm and the lateral leaflet movement of the Indian telegraph plant. We will than take a few of the numerous biological rhythms which evolved as adaptations to the 24 hour time structure of the earth and which are especially suited for experimentation. For instance the leaf- and petal movement and rhythmic fragrance in plants, the rhythmic change in shape of a marine amoeba, rhythmic events in animals and man. The importance of the 24 hour periodicity has in the last years been studied intensively in respect to shift work, flights crossing time zones, and in certain deseases . The influences are meanwhile well known in the public. Less well known is the participation of the 24 hour rhythmicity in the measurement of the daylength . In this way many organisms, especially from temperate and higher latitudes, are able to determine the time of the year reliably. This has been pointed out already in the thirties of the ninetheens century by Bünning. The field of photoperiodism has in the meantime grown considerably and is of much practical importance. There are also numerous interesting studies on tidal and lunar rhythms as adaptations of many marine organisms to the coastal conditions of the oceans. However, we will not propose experiments for this field in this book. For information, a few movies can be used at school (see page pageref).
The third part of this book is devoted to different ways of teaching the field of rhythms in organisms. Simple experiments without expensive equipment are suited for schools and the interested layman (see page pageref). Teaching aids, teaching proposals, and didactic considerations are found from page pageref onward. You will find also hints for getting, rearing and keeping experimental organisms, sources for obtaining equipment and laboratory material (page pageref).
As is usual in life, the best way to learn something is by doing.
Verba docent
exempla trahunt5
A few technical remarks: This book was for some years in the process of evolving. It was used by students who participated in a course covering the area of chronobiology. This year (1999) it was polished up. Especially most of the figures were improofed. For diagrams I used `Scandata� in order to obtain data from curves in figures from publications. They were produced with `Techplot�. Both programs are from Dittrich, Braunschweig. Vector graphics are made with `Killustrator� under the operation system `Linux�. Text was applied to bitmap figures with the same program. The book text was written with `Lyx�. It uses Latex in the background. This program was also used under `Linux�. The figures are inserted as eps- or ps-files and the book stored under ps- or pdf-format.
The formulation of a problem is often
more important then its solution
A. Einstein
Overview:
In scientific work certain rules are used which have proofen to be useful.
The method of multiple working hypotheses will be explained. We will learn how
to put forward and test critically hypotheses and how to plan, execute and analyse
experiments by using as an example the telegraph plant Desmodium motorium.
The significance of communication in science will be treated. Controversies
and their function in science will be mentioned briefly. Finally we will point
to some unsolved problems in the area of chronobiology to motivate you for own
studies.
Textbooks give often the wrong impression, that nature is well understood by man. They stress the collected body of knowledge and the problems solved, whereas the unknown is often not even mentioned. As soon as one starts to study more closely a special area of natural science it becomes, however, very obvious that many things are unknown, not studied or not understood.
In the area of chronobiology this is especially obvious. It is a relative young branch of biology and therefore not much knowledge has been accumulated. For the student this has the advantage, that he can do research work with simple methods and without intensive literature studies.

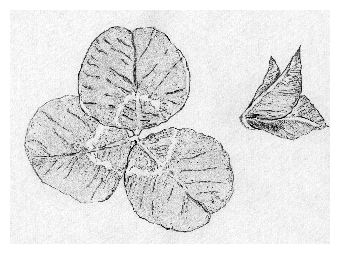
In this part we will get to know some general methods of scientific work. They can easily be transferred later to similar situations. Scientific work consists to a substantial part of answering questions. Questions arise, for instance, by observing. If we walk in the evening through a meadow, the threeparted leaves of clover are differently positioned as compared to the day time. They are folded together in a vertical position, whereas during the day they are spread more horizontal (figure 1.1). How does this leaf movement work? We try to find answers (so called hypotheses) and to test them experimentally.
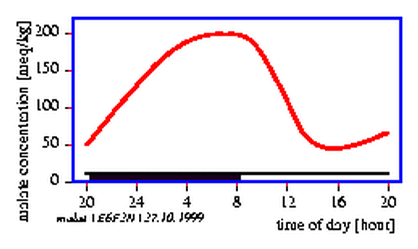
Or we observe in the Gentianaceae Exaccum affine an intensive fragrancy in early afternoon. In the morning and evening the odour is less intense (figure 1.2).

Golden hamsters are night active and rest during the day (figure 1.3). Is this a direct consequence of the light-dark-cycle? Or would the animals show a rhythmic change between activity and rest also under constant light conditions (e.g. under very weak light)?
Such observations lead often to questions, which can be answered tentatively by hypotheses and be tested by experiments:
phenomenon � problem � hypothesis � experimental test
According to [23] and [105] scientific work is favoured by the consequent use of the `method of multiple hypotheses' and `strong inference'. The method consists of the following steps:
The method of multiple working hypotheses was put forward in 1890 by Chamberlain, a geologist, in a lecture of a meeting of the Society of Western Naturalists. It avoids the danger of the method of simple working hypothesis, were one clinges with motherly affection to one owns hypothesis. This method is therefore especially suited for areas, in which much knowledge has accumulated, but much more is unknown.
Proposal: Read the reprint of the original article of Chamberlain `The method of multiple working hypotheses' [23]. Platt in 1964 has taken up this method and recommended it as especially useful for scientific work. He calls this strategy strong inference. The different steps have been mentioned already and we would advice strongly to read this article [105].
Using the way of scientific work in the `Laboratory of Molecular Biology' in Cambridge as an example, Platt demonstrates, how this method, if used systematically, leads rapidly to progress in solving problems. This is a problem oriented instead of a method oriented procedure. Each day the newest results are written on the blackboard, what went wrong, how errors can be avoided, and how the hypotheses can be tested critically by experiments. New experiments and controls to test the hypotheses are proposed. It is always advisable to use the simplest system, which does still show the characteristics one is interested in. It is more important to ask which experiment disproofs a hypothesis than to ask how to proof it:
'Verwende das einfachste System, das noch die Eigenschaften zeigt, die Du untersuchen willst'.
Scientific work is comparable with the work of a detective in solving a crime.
By observation or by oral or written information he is confronted with a problem.
He tries to solve the `case' by finding all kinds of possible explanations (`hypotheses').
Neither an able detective nor a good scientist would follow up just one single
hypothesis.
If you play the game `mastermind', where you have
to find out the sequence of differently coloured pins, it would be unwise to
begin with a fixed idea of the correct solution, before you have collected enough
informations. This game shows incidently, what is also true for science, that
`negative results', i. e. our guess was wrong, lead normally more rapidly to
a solution as compared to a partly correct result. If, for instance, in our
game out of four differently coloured pins none is correct, we know that only
one or two of the remaining colours had been set.
`Each conclusion, which is not an exclusion, is uncertain and must be tested again (Lederberg)'.
'A theory, which can not be vitally hit, can not live (Bacon)'.
'Hypotheses, which cannot be disproofed, are meaningless (Bacon)'.
Proposal: If you dont know the game `mastermind', play
it with somebody.
Proposal: Read the article by Popper `Von den Quellen unseres Wissens und unserer Unwissenheit' [106] and a detective story (DuMont's Criminal-Rätsel [133]). In this detective story the solution of the `case' is reserved in closed pages. Use the method of multiple hypotheses in trying to solve the case.
Besides putting forward hypotheses, important steps of scientific work are:
We will exercise these steps in different examples.
Using the drinking duck6 (figure 1.4), we will see how questions arise from observations and how they will lead to hypotheses.
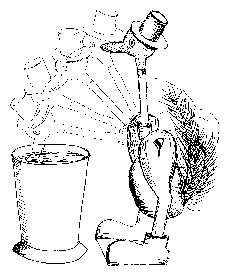
Instruction: Fill the beaker with tap water up to the rim and dip the beak of the drinking duck into the water, until the felt of the head is wet. Bring duck back in its original position and observe it. Note down your observations. What are the questions arising? Which hypotheses could answer your questions? How could you experimentally test your hypotheses? If the problem is too complicated, it usually helps to subdivide it into smaller problems. They are more easily solvable and might finally lead to the solution of the main problem.
The analysis and interpretation of data shall be demonstrated by using the rhythmic movement of the lateral leaflets of Desmodium motorium, the indian telegraph plant. This plant exhibits vertical movements of the small lateral leaflets (`telegraph plant'). Look at the movie `Desmodium motorium (Fabaceae) - Gyration' [127] and observe the movement on the plant directly (figure 1.5).
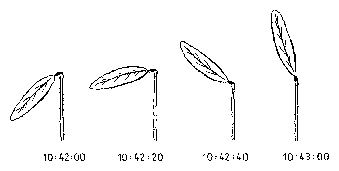
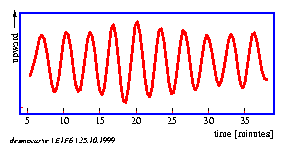
The oscillation is quite regular. We want to determine the duration of one cycle of the up- and down movement (figure 1.6). Sequential periods are not exactly the same. We have to measure therefore several cycles and to calculate a mean value of the period. We should also find a measure of the variability of the period. For recording the time we use a stop watch. What is the best way to determine period length?
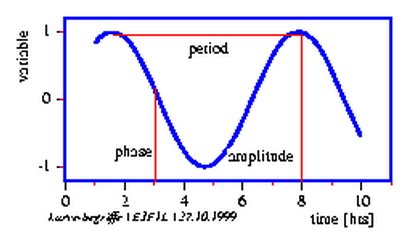
Period length is the time between identical phases of the oscillation:
for instance the time between a maximum and the following one, or between minima,
or between the point of inflections (figure 3.1). Which phase (=fixed
time in a cycle) would you use and why? We will become familiar in a special
chapter (page pageref) with methods in which period length is determined
by using all the recorded data.
|
|
x |
|
� |
|r|c|c|c|c|
Nr.
1
Why did we not determine the time between the first maximum, and the
last maximum and divided it by the number of oscillations? The reason is, that
we want to determine the variability of the period. In order to calculate the
variability, we need the individual period lengths. Variability is
caused by biological and methodological reasons. As a measure of the variability
we will use the standard deviation (there are other measures of variability).
|
|
x |
|
x |
With the help of the standard deviation we can find out which phase is the most
suitable one to determine the period length. It should have the lowest variability,
i. e. the smallest standard deviation. Find out whether the maximum, the minimum
or the point of inflection to the minimum or to the maximum is the most suitable
one (table 1.3).
| time | result | standard deviation | result |
| xmax = | SDmax = | ||
| xmin = | SDmMin = | ||
| xPI\nearrow = | SDPI\nearrow = | ||
| xPI\searrow = | SDPI\searrow = |
The standard deviation can also be used to test whether the mean is significantly different from another mean (if, for instance, the period length was determined at another temperature, see page pageref).
For this purpose the standard error SE is calculated from the standard
deviation SD using the following formula:
If the means are by 2 or
more standard errors apart from each other, the difference is highly significant.
SE = SD/�n
Planning of an experiment is one of the main tasks of a natural scientist. Here phantasy and creativity are needed as well as carefullness, exactness, wariness and critical proceeding. If we want to explain a problem or a phenomenon, we try to explain the occurrance or functioning: We put up one or more hypotheses. With an experiment we critically tests these hypotheses. In the following a few basic principles are listed [135].
How would you build a recording device to record the lateral leaflet movement rhythm of Desmodium motorium? A temperature controlled box is used, in which temperature can be kept constant at a certain value (see page pageref `Building an air conditioned box'). An electronic thermometer serves to measure the temperature.
Plan an experiment in which the dependency of period length of the leaflet movement
rhythm from the temperature is studied. Execute the experiment and use the analysis
method explained in the foregoing section to determine mean values, standard
deviation and standard error.
Plot the period length as a function of the temperature. Add the standard errors
as vertical bars to the means. Interprete the results and write a report of
your experiments (see page pageref). What is the Q10 -value?7 It is calculated from
|
Some hints and helps: For growing the plants see the section on the telegraph plant Desmodium motorium in the chapter `ultradian rhythms' on page pageref. For recording the movement you can use the setup shown in figure 1.7[82]. It is easily constructed. Or you use the digitizing method described on page pageref. Cut off with a razor blade a leaf with lateral leaflets which are moving well. Put it immediately in a small hole in a polyurethan disc floating on distilled water in a small vial. Transfer to the air conditioned box. If you use the Koukkari method, stick (with a tiny amount of water soluble glue) a delicate thread or a human hair to the tip of the leaflet and to the end of the delicate wire balance. Cut off the second lateral leaflet and the terminal leaflet. Add a small droplet of vaseline to the cut surface.
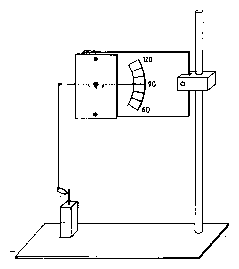
The measurement is better done by two persons: One calls the times at which the phase is reached which serves as the reference phase. The other person reads the time from a running stop watch and notes it down. Use the table 1.4 to enter the angles of a Desmodium motorium lateral leaflet every 15 seconds. It is the basis for a graphical display of the values as a function of time (figure 1.8).
| time | temperature( 0 C) | time | temperature( 0 C) | |||||
| sec | 150 | 250 | 350 | sec | 150 | 250 | 350 | |
| 0 | 540 | |||||||
| 15 | 555 | |||||||
| 30 | 570 | |||||||
| 45 | 585 | |||||||
| 60 | 600 | |||||||
| 75 | 615 | |||||||
| 90 | 630 | |||||||
| 105 | 645 | |||||||
| 120 | 660 | |||||||
| 135 | 675 | |||||||
| 150 | 690 | |||||||
| 165 | 705 | |||||||
| 180 | 720 | |||||||
| 195 | 735 | |||||||
| 210 | 750 | |||||||
| 225 | 765 | |||||||
| 240 | 780 | |||||||
| 255 | 795 | |||||||
| 270 | 810 | |||||||
| 285 | 825 | |||||||
| 300 | 840 | |||||||
| 315 | 855 | |||||||
| 330 | 870 | |||||||
| 345 | 885 | |||||||
| 360 | 900 | |||||||
| 375 | 915 | |||||||
| 390 | 930 | |||||||
| 405 | 945 | |||||||
| 420 | 960 | |||||||
| 435 | 975 | |||||||
| 450 | 990 | |||||||
| 465 | 1005 | |||||||
| 480 | 1020 | |||||||
| 495 | 1035 | |||||||
| 510 | 1050 | |||||||
| 525 | 1065 | |||||||
A written protocoll is an undispensible part of scientific reasearch work and certain rules have been found to be of much help (see e. g. [135], page 130ff.):

This section is mainly for students, for whom it is an important part of scientific work.
A scientist who has finished a research work usually reports the results. He would first explain it to his colleagues, who will constructively criticize methods and execution and propose alternatives to interprete the results. He would then give a talk to a larger group of scientists. This could be a colloquium of the department or a meeting (see the following invitation).
|
Kolloquium des Sonderforschungsbereichs
|
It might take a long time from finishing the manuscript until acceptance by a journal. Therefore often short communications are published before the final publication, which have less stringend demands. Interesting new developments and results are reported and commented on in special journals such as `Science News' and others.
How does such a publication look like, how is it structured and how does one read it?
Proposal: Read the publication of Sulzman et al.: `Neurospora rhythm in space: a reexamination of the endogenous-exogenous question' from the journal `Science' [125] from 1984.
Read afterward the paper of Mergenhagen in the journal `Die Naturwissenschaften' [92]. Mark the different sections according to the scheme in the preceeding paper. If you want a quick information about the content of the paper, the following procedure is proposed:
Title interesting? � Abstract � Figures � Introduction � Discussion � Result
First decide by reading the title, whether it is worthwhile for you to look
at the publication more closely. Then read the abstract or summary. If it is
of interest to you, look at the figures and tables and their legends, perhaps
also introduction and discussion. Important papers must of course be read
carefully also in the part containing the results. Sometimes one might be interested
in a method or a special point of the discussion only. In this case you would
start right away on the particular place.
The best method to find a good style in publishing a paper is to read scientific articles. In doing so you can learn from good and bad examples. There are also books on it such as Ebel et al. [81,33,121].
Task: Write a paper on the execution and evaluation of your experiments concerning the temperature dependency of the Desmodium-leaflet rhythm. Use the usual structure of an article. Make a sketch of the experimental setup, a table for the data and a graph showing the dependency of period length on temperature. Do not forget to include a list of references at the end of the article (see also literature search).
Before we plan a scientific research project we would like to know whether the solution of the problem has perhaps already been found and published. Or, if not, we would at least like to know, whether other people have thought of the problem and what they found out about the background of my studies. Six hours work in the library might save you 6 month work in the laboratory.
Encyclopedias are the most general and useful sources of information if you want to become familiar with a research field. There exist furthermore literature guides for certain fields with basic references, reviews, and journals with abstracts such as [124]. Handbooks are also useful to become familiar with a new field [8,].
With textbooks you can familiarize yourself with the background, with monographies you can become acquainted with details of certain fields. Subject catalogues of libraries, catalogues of books in bookstores [30] are helpful in this respect. To decide whether a certain book is interesting for you, you can consult reviews on books (`Reviews')[45,]. An abstract- und index-journal is `Biological Abstracts'[14]. Instructions are included in the halfmonthly editions.
Quite useful is also the `Science Citation Index' [115](figure 1.11): We know an important, but older paper of the subject we are interested in, and we want to find more recent literature. This publication indicates all authors citing this particular article as a reference in their publication. It is likely that such a paper is also concerned with the same or a similar topic. With the help of the `Science Citation Index' it is therefore possible to find more recent publications to a topic.
The most recent papers can be found in the latest issues of scientific journals. The following journals are specialized in the field of chronobiology (figure 1.12):

In the `Current Contents' the pages of contents of journals of the biosciences are reproduced and they contain an index of subjects and authors. In it one can find the wanted information quickly. Under circadian rhythms, for example, you find on page 229, 4th column in number 14, volume 34 (April 8, 1991):
| 127 | 57 |
| 127 | 41 |
The first number is the page in the particular issue of `Current Contents', the second number is the page of the corresponding article in the particular journal (in the case shown a paper by Queiroz-Claret and Queiroz). The `Current Contents' are also available on diskettes and can be used with a comfortable program allowing to search the literature for key words, authors, journals, and to produce files and printouts of the relevant literature.
It is often helpful if one knows names of scientists working in the particular field (see Science Citation Index). You can write to those people or ask them. But you should have gotten first a founded theoretical and general background in the field. Sometimes it is also advisable to browse through areas which are not directly connected with your field of interest. In this way you might, for instance, find solutions for methodological problems.
'Wise selection rather than all-inclusive coverage is the key to library work'.
If you are planning to start your own literature file, you could make use of
modern data bases and data base systems. They are available through the university
libraries and in the internet, but also from publishers. 9With their help it is easy to use reprint collections and to find papers of
certain authors, according to certain key words, or according to e.g. the year
of publication. Printed literature can be read with a scanner into a computer
and made maschine readable with an optical character recognition program (OCR).
Proposal: Use the different methods mentioned above to find the more important publications of the last two years for a chronobiological topic of your choice. Make a literature list.
The method of multiple hypotheses and 'strong inference' is not always used by a particular research worker, perhaps because he does not know it or because in a special case (where the problem is too complex to allow meaningful alternatives) the method is not useful. In these cases alternative hypotheses are put up at another level. Other scientists might disagree with the proposals and inferences and it comes to scientific controversies. One such controversy from the field of rhythm research is given in the following:
Proposal: Read the book of J.D.Palmer (1970): The biological clock: Two views. [101]. In this book two controversial views are discussed, how circadian rhythms arise. Brown denies, that they are the result of internal clocks of the organisms and claims, they are the direct result of external factors which are changing in a 24 hour pace. To exclude the light-dark-cycle or a temperature cycle, as is commonly done in research work on circadian rhythms, is according to Brown not sufficient. Instead, the rhythmic alternation of other factors should be prevented, which is, however, often impossible (magnetic fields, high altitude radiation etc.). Hastings, on the other hand, claims, that circadian rhythms are the expression of an internal clock inside the organism. It continues to run even under stringent constant conditions of the environment. This view is supported nowadays by most scientists working in the field. Describe the two views in your own words. How do the two authors explain freerun? Collect the arguments for the two hypotheses and note down the weak points of each. Try to find crucial experiments which can be used to test the hypotheses critically. Read in this context the following papers, which have been published after the book had appeared.[57,,,]
We have stressed already in the introduction, that many problems in the natural sciences are unsolved and that we are far from understanding the nature around us. One of the main goals of science is, to reduce ignorance and superstition of mankind.
Unsolved problems are numerous in the field of chronobiology. The highest priority in the list of unsolved problems is the search for the mechanism of biological rhythms. In no case this question has been answered satisfactorily at the physiological safe at the molecular level. A number of models exist [38,] which are, however, partly unsatisfactory or even wrong. New methods to clarify the mechanisms involved come from the field of molecular biology and systems analysis. Important in this respect are not only the methods, but also the systems studied. It is recommendable to use a minimal system, which shows the property to be studied, but exhibits only few phenomena which might disturb the studies.
In trying to unravel the mechanism of circadian rhythms a prokaryote would be more suitable as compared to a eukaryote: The former is much more primitive in respect to structure and function. The genetical structures are extremely simple, consisting of a ring-shaped chromosome without nucleus. Organelles and compartments are absent. New molecular genetic methods can be used [107].
Another minimal system are specialized eucaryotic cells such as the red blood cells of mammals. They are specialized to transfer O 2 . They do not possess a nucleic acid metabolism, have no mitochondria, no respiration, no protein synthesis. Finding a circadian rhythm in such a system would be an important step to understand the underlying mechanisms. A number of proposed models would be rejected if a circadian rhythm exists in red blood cells. And indeed a circadian rhythm of activities of different enzymes in red blood cells has been described [10]. However, attempts to repeat these results were unsuccessful in a number of laboratories [87,]. See also the preceeding subsection controversies in science (page pageref).
Finally, a circadian rhythm of dry seeds of beans was described [19]. This is another minimal system, since besides a very low respiration no other metabolic processes are found. This result has not been confirmed, although the experiments are simple and the results of paramount importance.
We have studied the lateral leaflet movement of Desmodium motorium and it would be interesting to understand the underlying mechanism. This is, however, a difficult problem and not solvable in the course of a laboratory exercise. In such a case it is advisable to divide the problem in smaller ones and try to clarify these. In the particular case of the lateral leaflet movement it would be recommendable to devide the problem into two questions: The question, what mechanism is responsible for the movement, and secondly, how the movement is controlled rhythmically. For the first question, the pulvinus and the turgor mechanism as the basis of the movement is of importance. For the second question of the timing, however, the kinetics of the movement, the temperature dependence of the rhythm, and the influence of inhibitors of various processes such as glycolysis or protein synthesis are of significance.
Proposal: Try to find a limited problem which can be solved during a laboratory course. Further unsolved problems can be found from the examples for rhythms in the second part of the book. For instance: which extraretinal photoreceptors are responsible for the synchronization of the locomotor activity of flies (see `Activity rhythms in animals', page pageref).
Overview:
Out of numerous recording methods we present two, which are quite versatile
and which have stand the praxis. One method serves to record movements and uses
a video camera connected to a computer via a frame grabber. With special programs
the pictures are analyzed. Furthermore an infrared light beam method is explained,
with which the locomotor activity of animals is recorded.
Many rhythmic events in plants, animals and also in unicellulars express themself as movements. They are easily observable. With a video camera coupled via a framegrabber to a computer and imaging methods such movements are recorded, displayed and analyzed.
The recording principle is shown in fig 2.1. The rhythmic leaf movement of a plant is used as an example. Images of the object are taken in predetermined sequences and via a framegrabber transferred to and stored by a computer. The position of a leaf is determined by a program which automatically recognizes the tip of the leaf. It saves the values of the position of the tip as x and y values. They can than be graphically displayed as a function of time. The period length of the oscillation can be determined by time series analysis programs.
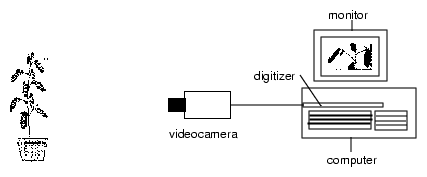
Since a handbook [37] exists for imaging, we will limit ourself here to the essentials. The video camera is connected via a cable to a framegrabber of a PC. (see also figure 2.1).
The imaging program needs Linux as the operation system. The programs are available on the internet and explained [37]. After starting the program, the object has to be put in focus (with the lense of the video camera) and the right size must be chosen (by adjusting the distance camera-object). A suitable illumination is important. To record in weak safelight or darkness infrared light is used. It can be produced with infrared light emitting diodes (LED). The object to be recorded should be clearly visible and not blocked partly by other structures. It is unimportant, whether the object is brighter or darker than the background. However, the lighting conditions should stay constant in time and space. Then the sampling rate of the digitizing and the recording interval have to be set.
Different kinds of data can be used for the analysis of the movement: The x- and/or y-coordinates of e. g. the center or the tip of the leaf, the number of pixels (as a measure for the size of the object). The parameter setting and a commentary can be stored in files. After a file name has been entered, the recording can be started.
During recording the data can be displayed on screen as a function of time.
The locomotor activity of many animals and mobile unicellulars is controlled by an endogenous clock. These movements can be recorded also by the imaging system just described. Examples are given in [36]. However, in the case of fast moving objects this system is not recommendable. We mention therefore an infrared lightbeam system which is described in a handbook [40] in detail.
Individual animals are kept in translucent dishes and supplied with water and food. An infrared light beam watches a part of the dish and transmits a signal, if the animal interrupts the light beam.
The system allows to record activity of up to 288 individual animals. It is fast (each animal will be monitored every 25 msec) and quite sensitive (it was originally developed for activity recording of Drosophila flies). It functions reliably also at varying light conditions and temperatures of the surrounding.
The activity of the animals can be displayed in form of an actogram: It is determined, whether in a certain time span (e. g. 4 minutes) the animal has interrupted the light beam for a certain number (selectable). Or, alternatively, it is recorded, how often the light beams are interrupted in a predetermined time span (e. g. one hour).
With the system the environmental conditions such as light intensity and temperature can be monitored. It is also possible to control temperature, light and other events with the system.
The data are stored in a peripheric processor unit (kind of minicomputer) via multiplexer (kind of inquiry mill, by which the data of different recording places pass the same data line) and are stored in a buffer connected to it. They are transferred hourly to the diskette of a host computer. A radio transmitter based clock serves as a time reference. The processor unit is buffered by a battery. In this way no data are lost in case of a power failure.
The data are available in a certain format, for which a plotting program exists. Furthermore the data can be transformed with a program (HELLRODA) into other formats. With special programs (see next chapter) the data can be analysed.
This system was constructed and the programs written by Winfried Hellrung10. The HELLRODA data transfer program was written by Joachim Schuster.
Overview:
If measurements of time variable parameters are performed
and the data stored, we obtain socalled time series. An overview on timeseries
analysis procedures and -programs is given. Using two examples from the field
of biological rhythms some of these procedures will be demonstrated.
If we want to study rhythms, we need not only to obtain data by using recording methods. We need also procedures to display and analyse the data. First elementary terms will be explained. Than the following questions will be dealt with:
This chapter is for students only, who need the informations for analysing experimental results.
Signals with a rhythmic component are characterized by at least three items:
With these parameters an oscillation can be characterized (see figure 3.1). Furthermore, the form of the oscillation is important: It can be sinus like or like a rectangle, a sawtooth or all kinds of intermediates. An oscillation might possess a trend (upward or downward) and can be noisy (figure 3.1). A timeseries could also consist of several oscillations which might superimpose each other.
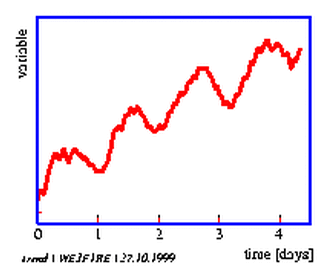
Almost always the first step in analysing a timeseries is the graphical display of the experimental results. In this way one can find out whether trends (oscillation does not deviate from a mean value, but tends to higher or to lower values) or noise superimposes the oscillation. The graphical display shows furthermore, whether one or more periodicities exist at all, which form the oscillation has, whether it is damped and whether the period is constant. The graphical display allows to decide, whether a timeseries analysis of the data is worthwhile. If the period length of the oscillation can be determined already from the graphical display, the following procedures are not always necessary, especially since they are time consuming. This is especially true for data which are not machine readable and have to be entered into the computer by hand, and if they are not available in regular time intervals (socalled nonequidistant sampling).
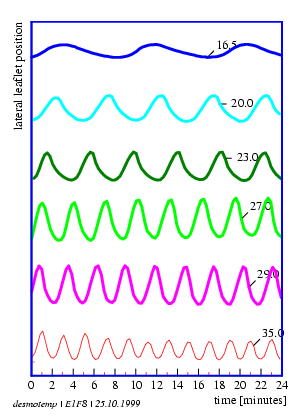
An example for the graphical display of a timeseries is shown in figure 1.6. The lateral leaflet movement of the indian telegraph plant Desmodium motorium was plotted as a function of time (see page pageref and legend for details). The period length is in the minute range. The oscillations are quite regular and the amplitudes large. The noise of the recorded data is small. Therefore the period length can be estimated directly without using special timeseries analysis procedures.
If several outputs are measured simultaneously as in figure 3.2,
the mutual phase relationship can be determined in a graphical way: The timeseries
is not plotted in the usual way as a function of time, but one output as the
function of the other output. The time information is still present, if for
each pair of data a dot is plotted in the coordinate system and if the dots
are subsequently entered as a function of time. With equidistant sampling the
distance of sequential dots correspond to a certain time intervall. If amplitude
and phase position of the two recorded outputs are stationary (i. e. do not
change with time), the later oscillations will lay on top of each other. If
they damp out, the curve turns into the center. If the periods are varying,
complicated figures will be seen.
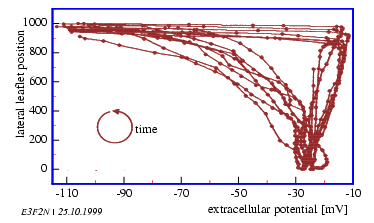
With the help of this kind of display the causal relation between recorded
values can be checked. In the example given it can be deduced, that the sum
potential of the pulvinus of the leaflet is closer to the oscillator as is the
leaflet movement, because the sudden hyperpolarization leads the downward leaflet
movement. It could furthermore be, that there is a causal relation between the
two items (which can, however, not be deduced from the plot).
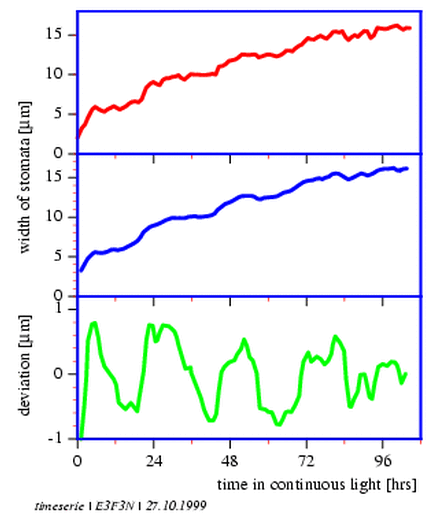
A close look at the upper curve in figure 3.3 reveils a steplike course. However, noise is quite pronounced. Therefore the values were first smoothened. For this purpose a smoothing average was performed with the values. Of five sequential values the average is formed and this value is stored instead of the third original value. The `smoothing window', consisting of five data, is now moved to a later record and the procedure repeated. In this way we get a mean value for the original fourth value. After all values have thus been averaged, the new timeseries is shortened by the two first and last values, but the deviations of the curve are reduced (center curve in figure 3.3). The smoothing improves with the length of the smoothing window (but at the same time more data are lost at the begin and end)12.
We see now the steplike course of the curve of the stomata data better (figure 3.3, center curve), but there is a strong upward trend.
A trend is usually disturbing for the analysis of an existing rhythm and should be removed by trend removal. After removal of the trend it can usually be better decided, whether a rhythm is pertinent.
A simple methode of trend removal is to fit the data with a so called polynomal curve. This fitted curve shows a minimal sum of the quadratic deviations from the smoothed individual values (a characteristic of a good fit). The deviations are shown in the lower curve of figure 3.3 and a rhythmic course is now clearly visible. Trend and noise are removed. The new data can now be analyzed for periodicities. How this is done will be described in the following.
The RUN-test is a simple and quick procedure to recognize periodicities. It is a distribution-free test, i. e. no special requirements are made in respect to the distribution of the data. After an eventually needed trend removal the mean value of all data of the timeseries is determined. Afterward the values which are larger then the mean are replaced by a + and the values which are smaller with - . In the next step the length of socalled `runs' is determined. A `run' is the sequence of + or - signs. If the timeseries is characterized by noise only, the lengths of the runs will be randomly distributed. If however a rhythm is present, the runs will fluctuate around the length of half the period.
For further analysis we use three procedures. They serve to determine the period length of a rhythm which has been shown to exist by using the RUN-test or by simple observation.
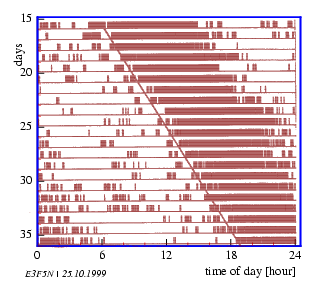
In this procedure the time series is cut into pieces which correspond roughly to the length of the period. The pieces are plotted underneath each other (see figure 3.4).
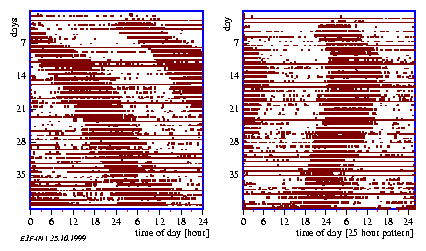
Usually one can now easily recognize whether corresponding phase points of the individual periods lie below each other: In this case the period length was correctly estimated. Otherwise the period was too short or too long. In this case the period can be estimated anew or a line is layed through the maximum of each period (see figure 3.5): Its slope reflects the period length. This procedure allows also to recognize changes in period length. In this case the periods can be determined for the different sections separately. This procedure is especially well suited for the determination of period lengths in actograms (see page pageref).
In the case of digital filtering (page pageref) the timeseries is transformed by a digital algorithm. In this way period lengths can be determined from rhythms which would be due to noise, otherwise less well analyzable (see figure 1.6).
The relatively new procedure of maximum-entropy-spectral-analysis (MESA) is especially well suited to determine even short timeseries reliably.
If the period length was determined, one might like to obtain an average curve of a period (see figure 3.6). For this purpose the signal-average-method of Cornelissen and DePrince [29] is useful. It superimposes the individual pieces corresponding to a period and determines the mean values with standard deviation. This method can also be used to analyse `residues'. If a periodicity was determined, other oscillations might still lurck in the data set which one obtains after the analysed oscillation was removed (`residues'). They can now be more easily analysed as compared to the situation where they were hidden by the main oscillation. This procedure is also useful for separating signals from noise.
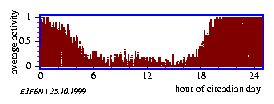
By measuring the locomotor activity of animals the distribution of events in time is obtained. It is usually displayed in the form of an actogram. The data are often reduced to yes/no events, i. e. the animal is active or inactive. To obtain an actogram, the timeseries is cut into 24 hour pieces und lined up underneath each other (see page pageref and figure 3.5). Similar to the frequency folding method these time spans can be chosen also in such a length, that they correspond to the period length (see figure 3.6 right part). If the corresponding values of the individual (subjective) days are vertically summed up, an `average day' is obtained. This average day can be compared with the corresponding average day of another time span which might have received another treatment (see figure 3.6 and the preceeding section).
With this method superimposed oscillations with close frequencies can be differentiated. With other methods this is difficult to do. For this purpose a frequency folding is performed with one period. The superimposed oscillation is now usually more easily recognizable from the figure.
A program package for the analysis and simulation of timeseries was developed by W. Martin in Bonn. It is available on large computers (e. g. VAX, IBM) and (sofar only) on a HP-computer. A detailed documentation is available [89]. The data must be equidistant.
The program allows in dialogues to enter and graphically display data sets (up to 9 timeseries at the same time). Two timeseries can be analysed simultaneously. The data can be manipulated (copied, deleted, supplemented, concattenated, selected, listed), characterized (maxima, minima, mean value, variance, standard deviation, standard error, variability, third moment, skewness) and analysed.
The analysis methods offered include complex demodulation (a band pass technique). Other analysis methods can be used such as periodogram analysis, spectral analysis, autocorrelation, Fourrier analysis, different filter techniques. Details are found in the description of the TIMESDIA program.
For literature concerning timeseries analysis see [89,,]. Programs for timeseries analysis are given on page pageref. Unfortunately for biologists there is still no useful book available for timeseries analysis. A collection of programs on disk for personal computers would be desirable.
Overview:
Models play an important role in the study of biological rhythms. They
allow to simulate the behaviour in time. The results of simulations are tested
with experiments and the model improved, if differences are found. We refer
here to some model programs and bring examples for rhythmic events.
If man wants to understand his surrounding, be it the physical, social or economic one, he uses models. This is, because the real systems are much too complex for analysis. Only by simplification we have the chance to understand it better. This holds also for rhythmic processes. Dynamical models are especially suited in this case. They consist of state variables (indicators of the state of the system), of combinations of state variables and of parameters of the environment which affect the system. If these parts of the system are known, the behaviour can be simulated [16].
To generate complex dynamical systems is quite elaborate. Fortunately procedure have been developed and programs are on the market which allow even the layman to construct dynamical models and to work with them. We will describe some of these programs and bring model examples from the field of biological rhythms with which oscillations can be simulated.
The `Deutsches Institut für Fernstudien (DIFF)' in Tübingen has developed the program `MODUS' for PC's13 [130]. With this program dynamical models can be constructed and used for simulations. Dynamical systems react time dependend. If their characteristic parameters are plotted against time, it is easy to recognize the behaviour of the system. In figure 4.1 an oscillating system is shown.
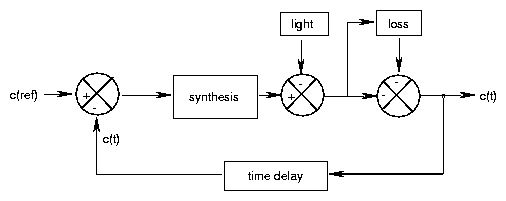
With Modus certain symbols can be connected with each other and used to represent dynamical systems as a graph on the computer-screen. The structur diagrams thus formed are then converted to corresponding equations by the program. Different methods exist. MODUS uses a notation originally going back to Hering et al. [63]. It is based on the system-dynamics method introduced by Forrester [50] and allows to describe interacting dynamical systems.
The following symbols are used by MODUS: State- and changing variables, functions, constants and connections (in the form of arrows). One can also construct model components which can be connected with each other. If you want to work with the model you should obtain the program and the detailed description from the publisher. In eight steps you are introduced into the handling of the program. On the program disks are also a number of examples, among others the predator-prey model describing oscillations between populations of predator and prey.
The DSP-program was developed by an audio-technician, M. Schick, and runs on the ATARI computer. Supply source see page pageref.
It allows simulations of digital audio-technique, control technique and non-learning neuronal networks. It can, however, also be used to build dynamical systems in biology, which oscillate. We add some examples in the appendix (page pageref).
You should get acquainted with the program by reading the instructions which come with the disk. In the creation mode, processes are created which are connected with each other as signal flows. Processes might have inputs and outputs. There are signal sources, signal transforming units and signal sinks (stores). Subsystems allow a hierarchical abstraction (block structure).
Matlab is a powerful program allowing also to simulate models.
We will get to know in the following some examples of models for biological rhythms. These examples are realizable with MODUS or with other programs.
Johnsson and Karlsson developed a feedback model which simulates for instance the gravitropic pendulum movement (see page pageref) [3], but also circadian phenomena [74]. It was used by Lewis [83] to simulate the circadian rhythm of locomotor activity of night active insects. The model consists of functional units which are shown together with their connections in figure 4.2. One can study with this model the influence of a changed environmental temperature, light-dark-cycle or different light intensities. In this way the reaction of the model to these environmental conditions can be checked and compared with the data obtained experimentally.
You might try the following situations:
Compare your results with those from the literature, which were found by Lewis using weta, a cricket from New Zealand [24,].
A slightly different feedback model simulates the lateral leaflet movement of Desmodium motorium. In contrast to the model explained before it shows a strong temperature dependency of the period. It is therefore useful to simulate rhythms as found in the lateral leaflet movement of Desmodium motorium. Try to determine the different period lengths at different environmental temperatures and calculate the Q10 value.
Further proposals:
Population rhythms occur, if a predator decimates the prey population. However, at a lower prey population density the predator has a reduced propagation rate. In the model of Lotka-Volterra these relations are described mathematically. It is assumed, that the death rate of the prey depends on the number of predators. The larger the predator population, the more prey animals will fall a victim. On the other hand, the predators are better off if many prey animals are available.
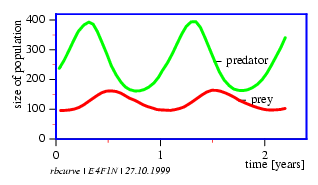
In the time diagram (figure 4.1) the number of predators and prey are plotted on the y axis. The x axis is a time scale with divisions in time units (generations).
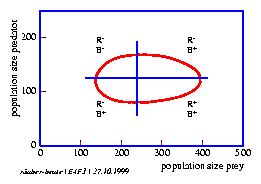
The Lotka-Volterra-model is also a feedback model: The prey population has a positive effect on the size of the predator population, whereas the latter has a negative (inhibiting) effect on the size of the prey population. Oscillations shown in figure 4.1 result. The predator population which so to speak growth on the prey population as a substrate, lags the prey population by 900 . This is shown in a phase diagram in which the number of predators is plotted on the y axis and the number of prey animals on the x axis. If the parameters are adequately chosen, both populations move on a closed path around the equilibrium point.
In nature such cycles were observed in snowhare and lynx in Canada. But things are more complicated than the model suggests, since it was found that the population of snowhare oscillates even in areas were the lynx has died out long ago.
A special model to simulate rhythms in animals was developed by Diez-Noguera for the PC14. With its help examples of rhythmic locomotor activity patterns were simulated in rats [31] and flies [61]. It uses coupled oscillators the properties of which are slightly different from each other. This situation is common in organisms.
We learned how to simulate different biological rhythms with different programs. The disk added to the book contains examples of models for oscillations which can be viewed at and changed with the different programs.
Overview:
Description of rhythmical events with periods in the minute and hour
-range. Examples are chemical reactions, reactions of seedlings after stimulation
by gravity, transpiration due to movements of stomata, and leaf movements brought
about by special joints.
Many biological oscillations are ultradian: The period length is shorter than circadian ones. Typically the period is in the minute- to hour- range. Chemical oscillations can serve as a model for this type of oscillations. The sequence of reactions in some of them are quite well known. They will be described in the next section.
In the third section of this chapter it will be shown how dark grown seedlings show pendulum-like oscillations after stimulation by gravity. Recording will be explained and experiments proposed.
The transpiration of grasses via their stomata can occur rhythmically, as reported in the fourth section. The water loss via the leaves is recorded and it is studied, how the oscillation depends on light intensity.
The lateral leaflets of the telegraph plant Desmodium motorium move relatively fast up- and down. These movements occur as the result of volume changes of motor cells in special joints. Recording of these movements and proposals for experiments are described in the fifth section.
As an example of a chemical oscillator the Belousov-Zhabotinsky-reaction is demonstrated. The temperature dependency of the period length should be determined. Besides oscillations in time this system exhibits also oscillations in space, which manifest themself as a pattern in chemical activity.
The rate of most chemical reactions depends on the temperature. We will therefore try to find out whether the period length of this chemical oscillator is also temperature dependent. We use the best known system, the Belousov-Zhabotinsky oscillator. This system shows also oscillations in space, which will be demonstrated.
For an introduction see [47,,]. The Belousov-Zhabotinsky-reaction was found by Belousov in 1958 and studied in detail by Zhabotinsky in 1964 [138].
According to the laws of thermodynamics all spontaneous chemical changes in a homogeneous and closed system are paralleled by a decrease in free enthalpy of this system. Oscillations would therefore be impossible. However, under certain conditions the concentrations of intermediates can oscillate around the expected values of the stationary state when approaching equilibrium (figure 5.1).
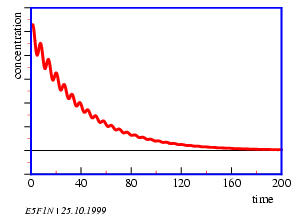
Oscillations in time and space occur. A prerequisite is, that the system is not yet in an equilibrium and that it contains a feedback. The Belousov-Zhabotinsky-reaction consists of two main reactions A and B. They interact only marginally, because the reactions of A consist of ions and molecules with paired electron spins (singlets), and B consists of reactions of radicals. Whether A or B dominates, depends on the concentration of the Br- ions: If the concentration of Br- ions is high, A dominates, if the concentration of Br- is low, B dominates. A uses up Br- ; therefore reaction B is induced. B produces Br- ; therefore reaction A is induced (figure 5.2).
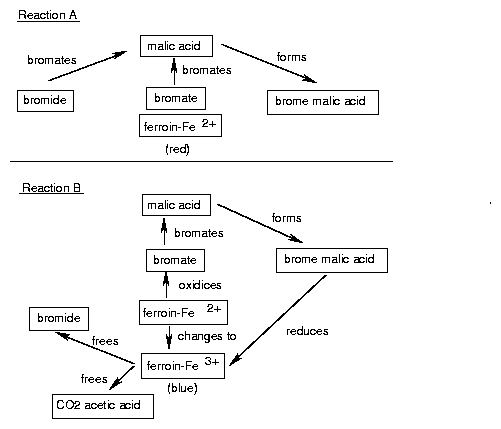
A: Bromide and bromate form together with malonic acid bromemalonic acid. The ferroin is first blue, due to the three valent iron (bromate oxidizes Fe2+ to Fe3+ ). However, due to bromate being removed, Fe2+ is formed and the ferroin turns red.
B: Brommalonate is concentrated enough to reduce Fe3+ to Fe2+ . Acetic acid, CO2 (gas bubbles!) and bromide are formed. The latter inhibits reaction A. This in turn inhibits the formation of brome malonate, and reaction B is inhibited. The cycle can start anew.
(A)(bromation of malonic acid)
2Br-+(BrO3)-+3H++3CH2(COOH)2� 3BrCH(COOH)2+H2O
(BrO3)-+4Ce3++CH2(COOH)2+5H+� 4Ce4++BrCH(COOH)2+3H2O
The rhythmic change in colour of the Belousov-Zhabotinsky oscillation is demonstrated in the following. The recipe of the necessary solutions are given and shown, how to observe the wave pattern of chemical activity. As an alternative, one of two movies can be shown which deal with chemical oscillators [64,].
The following four stock solutions are needed:
Pour the described solution in a clean plastic Petri dish (about 10 cm diameter) to a highth of 5 mm. A pattern of waves forms (figure 5.3). A trace of a detergent facilitates the wetting of the dish. The same chemical processes as described before are also responsible for the occurrance of these patterns. However, diffussion of the brome ions due to the lack of mixing plays now a role.

It should be determined, whether the period length (time between color changes from blue to red) depends on the temperature of the solution. This should help you to plan and execute a simple experiment. Therefore only some general proposals are made here and some hints are given, how to obtain the necessary informations. It is advisable to work through the chapter `Scientific work'.
Use the following procedure:
After having determined the period length at the different temperatures the mean period length and the standard error are plotted against temperature in a diagram. Read in the glossary about the Arrhenius plot and transform the values correspondingly. Determine the Q10 -value and interprete the results. For the graphic display you can use a plot program. What one should take into account in writing a report is described on page pageref.
The system oscillates only at temperatures up to 400 C. At this temperature the oscillation stops after 3 minutes. At room temperature the oscillations can be followed for about 4.5 hours. The concentrations should be kept accurately except the KBrO3 concentration (can be varied to twice the amount) and the Ferroin-concentration (can be varied eightfold). Traces of chloride ions (for instance at the fingers) can inhibit the reaction.
Dark grown seedlings may show a pendulum movement after stimulation by gravity. They will be studied using the morning glory (Pharbitis nil). The oscillations will be recorded with a video recording system. The period length and latency time is analyzed as a function of environmental temperature and hypocotyl length using different programs.
Gravity is used by many plants as a reference for orientation in space: Shoots grow often opposite to, main roots in the direction of the gravity vector. The physiological mechanisms of the balance system of plants are neither on a cellular nor on a molecular basis understood. The same is true for the balance system of man and animals. Experiments in space have offered new possibilities to study the function of the balance system of man in the absence of gravity. This is also the case for plants [134,,72,,].
Bending movements controlled by gravity are found in almost all higher plants. Cereals which have been layed down by wind grow upward again. Seedlings of sunflowers, morning glory and other plants which have been horizontally placed do the same. The physiological basis of this gravitropic reaction has been reviewed by Volkmann und Sievers [128] and by Iversen [68]. The sedimentation of amyloplasts (organells with starch grains) leads to an asymmetrical distribution and a stimulation of the endoplasmic reticulum. How this occurs is not yet understood in detail. According to Pickard [104], however, gravity is supposed to influence cell structures as a whole and induce stress reactions which lead to the gravitropic reaction.
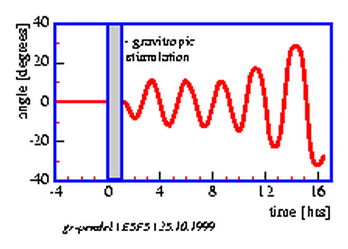
After gravitropic stimulation gradients of indolyl acetic acid (IAA), K+ and Ca2+ are formed [104,]. The intensity of the reaction depends on the strength and direction of the stimulus. Often the reaction to a geotropic stimulus occurs in form of a damped oscillation. There are, however, also endogenous, selfsustaining oscillations, e.g. in the case of pendulum movements. Such `circumnutations' of sunflower seedlings were modelled in 1967 by Israelsson and Johnsson [67]. According to the modell, the movement was completely dependent on gravity. However, a spacelab experiment in 1983 (spacelab 1) showed that under microgravity conditions oscillations were still occurring. They are, however, less regular [17]. Already Darwin [26] regarded circumnutations as autonomous movements, which occurred independent of external factors. A modell for the gravitropic pendulum must, therefore, assume the existence of an internal oscillator and an additional influence by gravity.
Seeds of the morning glory Pharbitis15 are treated for 45 minutes with concentrated H2SO4 in an Erlenmeyer flask and shaken once in a while (Attention! Danger of spatter). This treatment causes swelling of the seed husks. Pour acid in a sink filled with much water and add quickly much water to the seeds. The short, strong increase in temperature leads to a uniform germination of the seeds. Use running water over night to wash off the acid. Place a net on top of the beaker to prevent the seeds from floating off. On the next day the seeds are planted individually about 15 mm deep in sand in a plastic vial (2-3 cm diameter) and kept moist. At 30 - 320 C the seeds germinate after 20-30 hours. They are kept in green `safe light' (physiologically unaffective)16 until the hypocotyls are about 50-60 mm long. This takes at room temperature about 4 to 6 days.
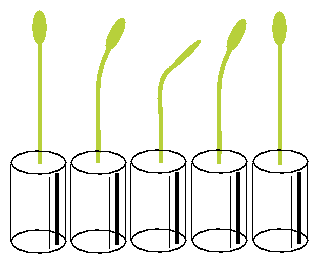
The vials with the seedlings are put into a horizontal position under safelight conditions for 20 minutes and brought back again in the vertical position. The seedlings start to bend after a latency period into the direction in which they would have grown if left horizontally. After some time bending in this direction comes to a halt, and the plants grow now in the opposite direction. Like a pendulum this oscillating movement continues for some time (see figure 5.4). The period length amounts to 20 minutes at 300 C [3].
Pictures of the hypocotyl are taken with a video camera and transferred to a computer equipped with a frame grabber. The position of the hypocotyl is determined by imaging analysis. The data are graphically displayed and analysed on the monitor screen (see figure 5.5). Function and use of the imaging system and the analysis programs are described in a handbook [37]. It is available on the internet [36]. The period length is determined by digital filtering. It has already been described in the first part.
Besides digital filtering other procedures can be used to analyse pendulum oscillations which have also been described in the first part of the book (periodogram analysis, maximum entropy spectral analysis, page pageref).
The transpiration of grasses via stomata may occur rhythmically. Water loss of the leaves is recorded and the dependency of the oscillation from the light intensity studied.
Land plants need CO2 from the air for photosynthesis. However, parallel to the uptake of CO2 water is lost, which might lead to difficulties in the water balance of the plant especially under dry conditions. Therefore during evolution special mechanisms were developed which help to solve the dilemma of a plant between hunger and thirst: A cuticula on the outer part of the epidermis cells which has a low permeability for water (and, unfortunately, at the same time for CO2 ), and special cell structures for the controlled uptake of CO2 and water transpiration, the stomata (figure 5.6).

The transpiration of plants via the stomata depends on the light conditions, the water status, the CO2 -concentration in the leaf tissue, on temperature and other conditions. Opening and closing of the stomata occurs often rhythmically and synchronously in the entire plant, although the external conditions are kept constant. This results in oscillations of transpiration and of the water uptake in the roots (figure 5.7).
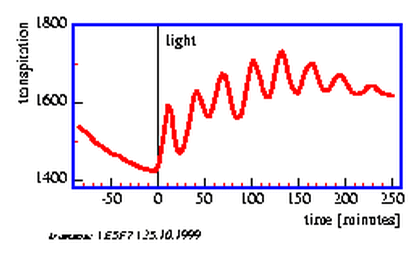
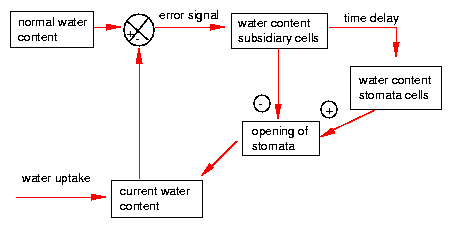
The control of the water state consists of several feedback loops. This allows oscillations to occur. Deviations from the normal water state change also the water conditions in the subsidiary cells and with some time delay in the guard cells. High water content of the guard cells induces opening, low water content closing of the stomata [25] [70](figure 5.8).
Ultradian rhythms in transpiration have been observed in grasses under certain conditions (see page pageref) [72][70]. To record transpiration we will use a humidity sensor. It monitors the humidity of the air after passing the oat seedling.
Rearing of oat seedlings: Oat grains are soaked over night on a sieve in a water container and brought to germinate. When the first leaf has ruptured the coleoptile (protecting sheath of the primary leaf) and has completely unfolded itself, a seedling is carefully removed with all its roots from the sieve. It is used for recording (figure 5.9).
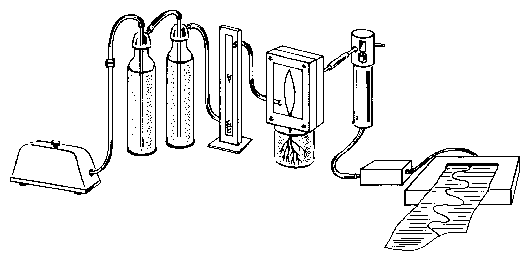
The leaf is mounted in a recording cuvette. Dry air is pumped through the cuvette with an aquarium pump. The air will be moistened by the plant to different degrees, depending on the amount of transpiration. The air passes a tube and reaches the humidity sensor. The electrical signal of the sensor is proportional to the humidity. It is amplified and recorded with a voltage recorder (figure 5.9). Alternatively the data can be brought directly to a computer via an A/D converter, stored and analyzed.
The cuvette is made of plexiglass as shown in figure 5.9. It consists of a front and back part, an entrance and exit tube for the air, and a small slit in the lower portion where the leaf is sealed with a mold17.
The humidity is measured with a humidity sensor (Vaisala, Finnland, see page 13.1.3). It consists of a condensor with a dielectric polymer between the condensor plates which is sensitive to humidity. Changes in humidity change the dielectric constant and therefore also the voltage between the condensor plates.
The cuvette to record transpiration is fixed to a support. The primary leaf of the plant is carefully inserted into the slit of the cuvette which had been filled with a mold. It seals the plant stem tightly from the outer air. The front cover of the cuvette is screwed to the back part with 4 bolds (see figure 5.9). Air is pumped through the cuvette by an aquarium pump via tubes through washing bottles. They are filled with dry (blue) silica gel. A flow meter just before the cuvette controls the air flow. Leaving the cuvette the air reaches the humidity sensor via a tube. The stalk of the sensor contains a converter, which produces an electrical signal. This is amplified and recorded by a voltage recorder. The plant in the cuvette is illuminated by a slide projector. Changing the distance and/or inserting grey filters allows to adjust the light intensity.
Getting started for recording
Setup of recorder
In order to analyse the data by digital filtering or another time series analysis procedure, the data have to be entered by hand from the recording paper to a computer. A more convenient way is to digitize the data with an A/D converter and to transfer the data to a computer. The curve can also be scanned and converted into data with a special program (e.g. Scandata). Plot programs (e.g. Techplot) are used to display the data as a function of time and time series analysis programs to determine the period length.
Alternatively, the position of the pointer of a voltage recording instrument is determined by imaging [37] as a function of time.
Besides the digital filter procedure other programs are available for the analysis of the transpiration rhythm. They have been described already in the first part of the book (page pageref).
The telegraph plant Desmodium motorium shows a fast up- and down-movement of the lateral leaflets. They are based on volume changes of motor cells in special joins. This rhythm is recorded with an imaging system .
Most people are not aware of the fact, that quite a number of higher plants display active movements. If corn has been laid down by wind or rain, it soon begins to grow upward again. But leaves can move too, as can easily be seen in Leguminosae: bean plants, peas, clover and Robinia bring during the night the leaves or leaflets in a vertical position, during the day they are horizontal. The movement is too slow to be recognized at once. But comparing plants in the day and night position shows the difference very clearly. In some plants much faster movements are found which are more obvious. The best known example is the rapid closing of the leaflets of the sensitive plant Mimosa. Not less obvious, but less well known is the up- and down movement of the lateral leaflets of Desmodium motorium, the Indian telegraph plant.
The lateral leaflets of Desmodium motorium show vertical or circular movements. This plant is therefore called `Telegraph plant' in India, because the leaflets seem to send messages between each other. Watch this movement carefully (figure 1.5). These movements will be used to practice the analysis and interpretation of data. We will record this movement with an imaging system (see page pageref). To analyse the data we use a program for digital filtering (see page pageref). How such a curve looks like is shown in figure 1.6.
Lithium ions slow down many circadian rhythms. We try to find out whether this applies also to the ultradian rhythm of Desmodium motorium. How would you carry out the experiment? Which concentration would you use, how long would you record, what kind of controls would you run?
Overview:
Circadian (daily) rhythms are widespread among higher plants. As an example
we will study the leaflet movements of Oxalis regnellii and the petal
movements of Kalanchoe blossfeldiana. The movements are recorded with
a video camera and a computer. Period length can be determined using special
analysis programs. Other rhythmic events in plants (fragrancy rhythm, acid metabolism)
are mentioned.
One of the most obvious daily rhythms in plants was described already by Androsthenes on the march to India with Alexander the Great: The leaflets of Tamarindus indica are folded together during the night and spread out horizontally during the day. These nyctinastic movements are widespread not only among the Fabaceae, but also in other plant families such as Oxalidaceae and Maranthaceae. The movements are brought about by turgor changes in special joints called pulvini (singular: pulvinus) . In other cases such as tomatoes and other Solanaceae or in cotton, Arabidopsis, movement is caused by alternating growth of the upper and lower part of the leaves [58,]. In Madurai (South India) 62 plants of a small botanical garden have been observed to show leaf movements (Chandrashekaran, unpublished).
There are several hypotheses trying to explain the significance of these nyctinastic leaf movements. According to Darwin [27] the heat loss to the surrounding is reduced in cool nights, if the leaves are in a vertical rather then a horizontal position (see also [43]). According to Bünning and Moser [] the vertical position reduces the amount of moon light absorbed. This prevents the plant from reacting photoperiodically at the wrong time of the year (for the significance of photoperiodism see page pageref). According to Karve et al. [76] the Fabaceae are able to grow very close together because of their nitrogen fixation capability. However, the resulting dense canopy of the leaves would not allow sufficient red light to reach the axels of the leaves in the evening and morning. This is, however, necessary for the induction of flowers. The vertical position of the leaves in the morning and evening would solve this problem.
Some flowers show also daily movements, such as the Crassulacean plant Kalanchoe blossfeldiana from Madagaskar (figure 6.1). Recording and experiments are described in this chapter. Other flowers show a conspicious fragrancy rhythm, such as Cestrum nocturnum (strongest odour during the night) or Exaccum affine (strongest odour in the early afternoon) (figure 1.2) [2,,].
The metabolism of plants is also often controlled by a circadian clock. The CAM (Crassulacean Acid Metabolism) in Crassulacea acidifies the cell vacuoles during the night and alkalizes it during the day (figure 6.2) [79].
Under constant conditions these rhythms continue. However, the rhythm is now not any longer exactly 24 hours, but circa 24 hours, in the case of Kalanchoe for instance 22 hours. This is a clear indication, that these periodic events are controlled by a circadian clock. Such movements are easily observable and we will use again for automatic recording an imaging system (figure 2.1).
These rhythms can be influenced by light-, temperature-, and chemical pulses. From the results of these experiments conclusions can be drawn concerning the underlying mechanism.
To demonstrate circadian leaf movements we use the leaves of the south American wood sorrel Oxalis regnellii. The threeparted leaflets are folded together during the night and are downward directed. During the day they are spread out horizontally (figure 6.3).
The plants are kept in many botanical gardens and easy to rear. The vegetative propagation from rhizom bulbs is the easiest. Put a bulb in a broad flower pot containing peath rich soil and keep well watered. The light intensity should not be too high. In case of diseases it helps to cut off all leaves. The plants start soon to develop new leaves.
The leaves are cut off the plant and the petiole put in a plastic straw which is mounted in the center of a polyurethan disk floating on water in a cuvette (figure 6.3).
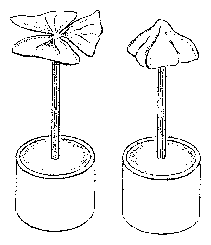
With a video camera the cuvettes are recorded from above. The camera is connected via a framegrabber to a computer. With imaging software as described before (page pageref) the movements can be recorded and analysed. The setup for the recording is shown in figure 6.4. For illumination green fluorescence tubes (Philips TL 20W/15) are used. The optimal light intensity is 1.4 Wm-2 .
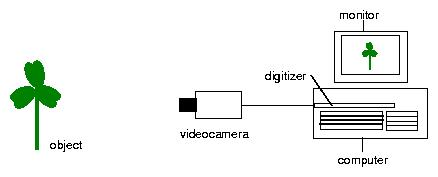
The data are displayed as a time series with a plot program (page pageref). Period length is determined by digital filtering or other analysis methods (page pageref).
A characteristic of circadian rhythms is that the periodic changes continue even under constant conditions. We record such a `freerun' (see next section). To understand the anatomical basis of leaf movements we make cross-sections through the pulvini and study them microscopically.
The leaves are cut at the end of a light-dark-cycle and mounted in cuvettes. They are then transferred into a recording chamber and exposed to continuous green light. Recording with an imaging system is started and run for 7 to 10 days. Every 20 minutes an image is taken.
The period lengths are determined by using adequate programs. Under continuous light the period length should not be exactly 24 hours anymore as was the case in a light-dark-cycle, but deviate from it. This proofs the circadian nature of this oscillation.
Cross sections through a pulvinus shows the structure and position of the motor cells (cells with large volumes) between the epidermis and the central core of phloem, xylem and strengthening tissue. In the leaf stalks, on the other hand, the conducting vessels and strengthening tissue are more at the periphery. Such a structure would not alow movement (figure 6.5).
You could study other plants with rhythmic leaf movements.
Proposals: Check different species of clover, bean, wood sorrel, Maranthaceae.
The petals of the flowers of the Crassulacea Kalanchoe blossfeldiana exhibit a diurnal movement. They are open during the day and closed during the night (figure 6.1).
This rhythmic movement continues even during constant conditions of temperature and continuous weak green light. The time span from one maximal opening to the next is now, however, only 22 hours and not 24 hours. This movement is the result of a volume change of the upper epidermis and mesophyll cells (figure 6.6), which in itself is caused by changes in the osmotic value of the cell vacuoles (figure 6.7).
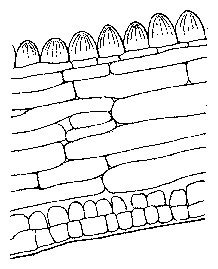
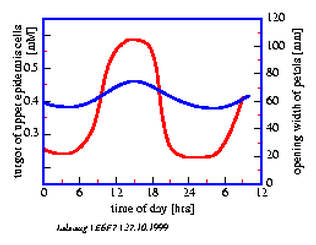
Plants are induced to flower by short day treatment (11 hours light, 13 hours darkness per day) for some months. After the flowers are formed and have started to open and close daily, they are transfered into a 12:12 hour light-dark-cycle (end of light for instance 10:00 in the morning) at e. g. 220 C. We record with an imaging system (page pageref). The flowers are broken off with a pair of forceps from the plant shortly before the end of the light period (in our case 10:00 o'clock) and mounted in a polyurethan plate (figure 6.8). The plate floats on top of a 0.2 molar succhrose solution in a rectangular plastic dish19.
The dishes with the flowers are illuminated from above by two green fluorescence tubes (Philips TL 40W/15). The tubes are covered with a green coloured foil (Cinemoid Foil Nr. 39, Strand Electric) and two grey foils (Cinemoid Folie Nr. 60). The light intensity at the level of the petals should be about 35 lux, because at higher intensities the rhythm of the petals is damped.
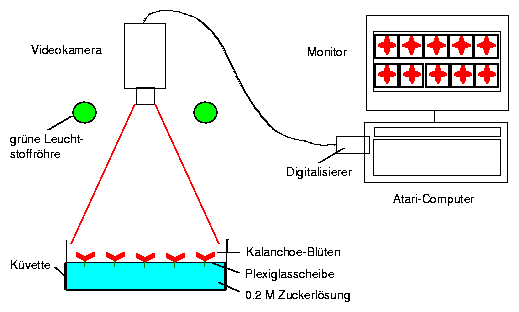
The imaging program is started on the computer. It is sufficient to sample images every hour for 7 days. Add daily some distilled water to the cuvettes to replace the lost water.
To analyse the results a time series analysis program such as digital filtering is used. It has been described before (page pageref). It filters the sampled data digitally and determines the period lengths of the petal movement rhythm of each individual flower (figure 6.9).
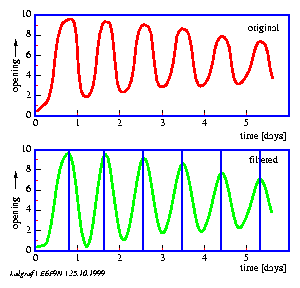
Calculate the mean of the periods of the individual flowers of the different groups and plot the results graphically.
Three experiments are proposed: In the first two the effect of Li+ -ions and of TEA (tetraethylammonium chloride, a potassium channel blocker) on the period length of the petal movement rhythm is studied. In the third experiment the turgor changes in the motor cells of the petals are determined. Turgor changes are the basis of the petal movement.
Kalanchoe-flowers can be used to study the effect of substances on the circadian rhythm. In this way the underlying mechanism might be better understood. A 5mM Li+ -solution for instance slows the clock by about 2 hours. Use 1 und 5 mM and a control, which contains only sugar solution. Li+ might work via the phosphoinositol-cycle.
Perform the experiment and evaluate it. Write a report of the results by using your protocoll (see page pageref).
During the leaf movement transport of ions through cell membranes plays an important role and K+ -channels are participating. Tetraethylammoniumchlorid (TEA) inhibits K+ -channels and can therefore be used to check, whether such channels are indeed parts of the rhythmic petal movement of Kalanchoe.
Use a control group (first cuvette) and three experimental groups with 0.5, 1 and 5 millimolar concentrations of TEA in 0.2 molar saccharose solution. Record for 7 days and analyse the data as described already. Plot the period lengths against the different concentrations used. Compare also other characteristics of the rhythmic movement such as damping and the opening width at the end of the experiment. What are your conclusions about the possible effect of TEA?
Read the article of Satter et al. [113], Ruge and Hampp [111] and Mayer and Hampp [91] on the basics and hypotheses of sleep movements in plants.
The red coloured upper epidermis of petals is peeled off with a fine pair of forceps. It is transferred into a mannitol solution (0.3 to 0.5 molar) on a microscope slide and covered with a cover slip. After 2 to 5 minutes the number of plasmolysed cells is determined under the microscope. This is repeated every 3 hours with epidermis pieces of other flowers. To avoid night work you can keep some of the plants 1 to 2 weeks before under an inverted 12:12 hour light-dark cycle. The 12 hour light period is during the night and darkness prevails during the day. In this way epidermis pieces of the normal and inverted groups which are observed for measurements are at each time physiologically 12 hours apart.
Quite a number of plants show a fragrancy rhythm of their flowers [2,,]. This is often an adaptation to the pollination by insects. Study different plants which are in blossom and try to find out how one can check for a rhythmic sensitivity of the human nose.
Overview:
Circadian rhythms are not only found in multicellular organisms, but also
in unicellulars and in cells of tissue and organs. Circadian rhythms have been
described lately even in prokaryots (in cyanobacteria). As an example for rhythms
in unicellulars the somewhat special case of Thalassomyxa australis is
presented. Here, in contrast to the normal circadian rhythms, the period length
depends on temperature and synchronization by zeitgeber is unusual.
The marine naked amoeba Thalassomyxa australis was detected by Grell in 1983 at the west coast of Australia [55]. It changes its form between a round, inactive phase and a moving phase, in which plasmodial extrusions are formed, by which the animal moves around and takes up food (see movie [54]). The change between the inactive phase r and the active phase a occurs rhythmically and the period length depends, in contrast to normal circadian rhythms, on the temperature [122]. At temperatures of 20 0 C the animals spend about 2/3 of the time in the resting phase and 1/3 in the active phase. The period length is about 28 hours. As food organisms unicellular marine algae can be used. Amphiprora, the green alga Dunaliella and a marine Chlorella species are useful.
Look at the movie `The change in form of Thalassomyxa australis'. At the end of the movie a scene is shown in which concentric bands are formed. Play back this part and look at it again. Try to understand how these rings are formed and write down your ideas. How could one determine the time between two neighbouring rings of deposition? Do you know other rhythmic events which manifest themself in a similar way? Comment on it.
Thalassomyxa and food organisms are kept at the department of zoology at the university of Tübingen. The amoebae are reared in seawater at 20 0 C and in weak light in Petri dishes. Diatoms (Amphiprora) serve as food. Usually the diatoms divide fast enough and only rarely they have to be added to the culture. The cultures can, however, die out after some time. Renew the seawater in time or transfer the amoeba to new dishes by putting microscope slides in the dishes. Once the amoebae have settled to the new slides, they can be transferred to new dishes.
The animals can be observed under the microscope at 100* magnification. The inactive phase is readily distinguished from the active phase. If the dishes are checked every 2 to 3 hours, the changes in form can be recorded as a function of time (figure 7.2). With an imaging system ([36,]) the process can be recorded and reproduced as a time lapse movie.
Four experiments are proposed. They are described in the following. Use the hints that are given earlier in this book on how to plan experiments and to write protocols for experiments.
To synchronize biological clocks with the environment, `Zeitgeber' (time giver, time cues) are used. The most important Zeitgeber to synchronize circadian rhythms is the daily 24 hour change of light and dark. How would you try to find out whether the change in form in Thalassomyxa australis is synchronized by the light-dark-cycle? Do the experiment, evaluate the data and write a report (see page pageref). Which kind of other Zeitgeber could synchronize Thalassomyxa australis? How would you test your hypotheses?
Circadian rhythms as well as common clocks run at different temperatures of the environment at the same speed. Organisms in very constant environmental conditions such as some parts of the tropics or the sea with uniform water temperatures do not need to have temperature independent clocks. However, even under these circumstances the dependency of period length from temperature is low, although not quite as low as in organisms from higher latitudes of the earth with more pronounced daily and annual changes in temperature. Ultradian rhythms (period length in minute and hour-range), on the other hand, are usually strongly dependent on temperature (see the chapter Ultradian rhythms, page pagerefff.). There are, however, also temperature compensated ultradian rhythms known. How would you test whether the change of form in Thalassomyxa australis is, as other circadian rhythms, independent of the environmental temperature? Plan an experiment, execute it and analyse the results. Write a report (see page pageref).
If you want to study the biochemical and physiological processes during the change in form of Thalassomyxa australis, a method is needed to synchronize amoebae mutually. The first study has shown, that a light-dark-cycle does not synchronize the culture. There is, however, a very simple method to produce a synchronous culture. It is done by selecting amoebae in the same phase , which than stay in phase for many days because their period length is very similar (or because they mutually synchronize each other?20). The following method is recommend: Pour seawater out of a densely populated dish, drop a jet of seawater out of a pipette from 4 cm hights onto the amoebae on the bottom of the dish by spiraling the pipette over the whole dish. Shake vividly and pour all amoebae out which have lost contact to the surface of the dish (figure 7.1).
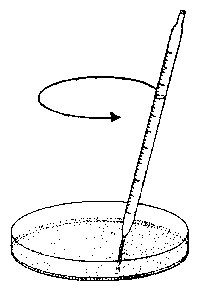
The animals which were washed off are the once in active phase. The animals in the resting phase are not detached from the dish by this treatment. Control under the microscope whether indeed all animals were washed off and repeat this procedure, if necessary. Add seawater to the dish, wait some hours and repeat the treatment. The amoebae collected then are all in a very similar phase, since they have gone from the resting phase into the active phase almost at the same time. They can be used as a synchronous culture .
If the collection of amoebae of the same phase is repeated after 12 hours, one can use the two cultures to test whether the cells are mutually influencing each other. Plan an experiment and the necessary evaluation, conduct it and write a report (see page pageref).
One can run also a computer model which simulates such a mixing experiment of amoebae in different phases with different numbers of amoebae at the begin. It is assumed that there is no mutual influence. An advantage of such a simulation is to get in a short time results of such different `experiments'. One can then look for those results which allow a clear decision. For instance, the optimal phase of the two cultures to be mixed and the number of cells in each can be found in this way. Intuitively one would probably mix a culture with a second one which is phase shifted by 1800 and would expect a new oscillation which results from the superposition of the oscillations of the two original cultures, and one would probably take equal amounts of amoebae from both cultures. If there is, however, a mutual influence of the two cultures, this combination is not recommendable, because the influence of culture 1 on 2 is the same as vice versa. It would be better to use a smaller number in e.g. culture 2 and to use a phase which differs from the phase of cultureone by 900 , because then culture 2 would after some time lock to culture 1.
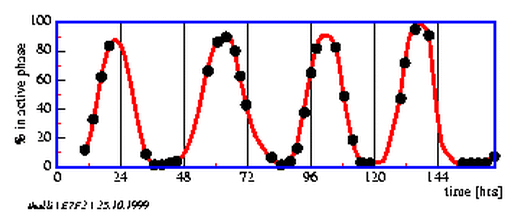
Overview:
In this chapter locomotor activity rhythms are presented which are wide spread
among animals. They will be described with examples of house flies, fruitflies
(Drosophila), and syrian hamsters
. Recording methods
will be presented.
The eclosion rhythm of fruitflies out of the pupal case serves as an example for a rhythm observable only in a population.
Finally we will get to know the circadian rhythms of man and their practical significance.
The behaviour of most animals is controled in a diurnal way. Thus, flies are active during the day and rest at night, whereas it is the other way round in syrian hamsters. Drosophila pseudoobscura flies eclose mainly in the early morning hours out of their pupal case. In the late morning only few emerge and in the evening and night none. Those rhythms are not only widely found, but also easily recorded. We will use again an imaging system for recording (see pageref).
First we will mention briefly the background of circadian activity rhythms. Afterward we describe how to get or rear the animals needed and how to maintain them.
Secondly we describe, how the different activities are recorded and analysed. We will use the methods described already in the first part of the book (imaging system und light beam method).
On page pageref the construction of an air condition box is described. It can be used to keep temperature and illumination constant.
Finally different experiments are proposed. We refer to a handbook in which the recording methods are described in detail and for which programs and data are available which help to understand the procedures better [36]. With this book comes also a collection of activity data which can help to learn the analysis and the understanding of actograms.
Locomotor activity of many animals is adapted to the light dark cycle. Night active animals rest during the day, day active animals during the night. Examples for the former are cockroaches, owls and mice, examples for the latter flies, many birds, lizards (figure 8.1).

This daily change in activity and rest is continued in a room with continuous light and constant temperature and food and water available ad libidum. It looks like the animals are still able to notice the change between day and night. This could be explained in two ways. Either one or more environmental factors are still synchronizing the animals which we haven't been able to eliminate, or the animals use an internal clock which tells when to be active or when to rest. It is easy to decide between both possibilities. In the case of synchronization by an external (unknown) factor the rhythm of the animal should continue to be exactly 24 hours, whereas an internal clock is unlikely to run with exactly 24 hours. The observation of our animals shows: whereas in a 24 hour day with light dark cycles the animals became active or tired about the same time, they were, under conditions without time cues, each day somewhat earlier or later active, depending on the speed of their internal clock. From this we conclude, that the internal day of an animal is circa 24 hours (`circadian' from latin circa, about, and dies, day), although very precise for each individuum.
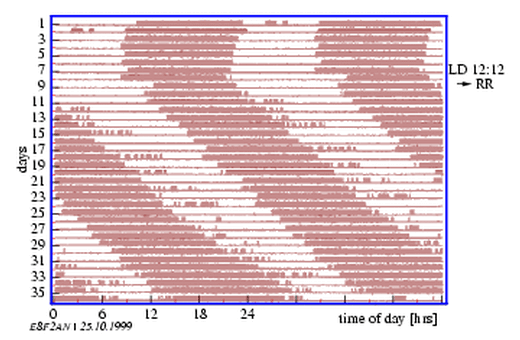
In figure 8.2 the locomotor activity of a housefly is shown. The animal was for 10 days in a light dark cycle and for another 30 days under constant conditions of weak red light and an environmental temperature of 22 0 throughout. This figure 8.2 is a so called actogram , a kind of black box of the fly (as used in trucks and air planes to trace the `activity' of the car or plain, respectively ): Activity is represented by vertical dashes, rest by a horizontal line. Time of day is shown on the vertical axis (from midnight to midnight), sequential days below. During the time of light dark cycles the animal became active with onset of light and stopped activity with the begin of darkness. When the change of light and darkness was discontinued, periods of high activity were still alternating with periods of rest. However, activity (and rest) began each day about 35 minutes later and stopped 35 minutes later. The day of this particular fly was therefore 24 hours and 35 minutes.
Musca domestica can easily be reared. However, since we need only a few flies and since they are usually found throughout the year in buildings, we will just give a reference only for rearing [131]. The identification of the species should be not difficult (figure 8.3) .

Drosophila melanogaster can be obtained from genetics departments or
purchased. During the summer and fall months they can easily be caught on fermenting
fruits
. Cultures can be reared
on food
which is prepared in the following way:
Measure
in the given order and stir when pooring the items into the warm water to prevent clumping. Boil for 5 minutes under constant stirring and add
poor while still hot in glass or plastic vials, close with foam stoppers and wait until cool. This food can be kept in the refrigerator for some weeks. A little bit of dry yeast should be added before flies are added [131]. Adult animals are transferred via a vial. They should have the same diameter of the opening. If the walls of the vial with food are wet, they should be dried with filter paper and/or a piece of filter paper should be put on the surface of the food before introducing the animals. Mark the vials (species, mutation, date) and rear the next generation in a 12:12 h light dark cycle at about 250 C. Larvae eclose from the eggs and feed on yeast growing in the food. Pupation occurs about 14 days later on the wall of the vials or on dry parts of the food.
Syrian hamsters can be purchased in zoo shops. There one gets also hamster cages which are already equipped with running wheels. Special hamster food can be bought, but corn or kitchen garbage are also sufficient.
Locomotor activity of animals can be recorded by different methods. Running wheels , tilting cages and photoelectric methods , in which the animal lives in a small container and interrupts a light beam when walking around, are examples. Here we use an imaging procedure for the recording of the locomotor activity of houseflies. This system has been mentioned already in the first part of this book2.1. The position of the animal in the cage is found out: Is it after a certain time interval still at the same place or has it moved? This recording is repeated for instance every 4 minutes and continued for several days. The results can be displayed on screen as an `actogram' and observed during recording. The data are stored in a file for later analysis.
For the recording of the locomotor activity of Drosophila-flies we use the system described in the first part of the book (page pageref).
With the locomotor activity of houseflies as an example the setup of the recording system will be demonstrated (figure 8.4).
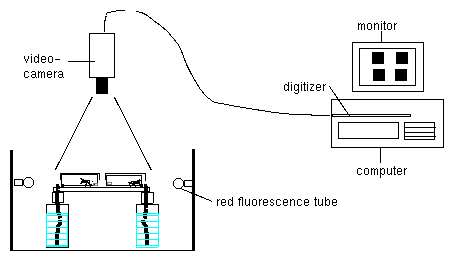
A hole (5mm diameter) is drilled in the bottom of a Petri dish of 10 cm diameter, through which a fly, head first, can be transferred into the container. A wick made of a strip of a kitchen cloth is inserted into the hole in such a way that it sucks water from a container and allows the fly to drink. A lump of sugar is fixed with plasticine to the Petri dish and serves as food. Several such cages are placed on a diffusing glass plate on top of red homogenous light22 (figure 8.5. The hole system is placed in the air conditioned box described already (page pageref).
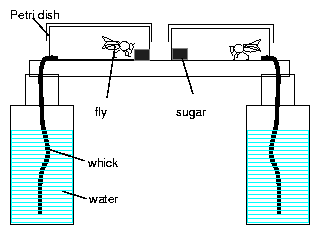
The video camera is mounted on top of the air conditioned box. The camera lens is situated in a hole and looks upon the dishes with flies, which are housed in the box on top of the red light. A cable connects the camera with the framegrabber, which is inserted into a slot of the computer. For details see [36,].
For the time span between the single pictures (sampling rate) we could choose for instance 4 minutes, for the recording time e. g. 7 days.
With an analysis program described in part 1 the activity data obtained with the setup can be displayed as an actogram (figure 8.2) and the period length can be determined (page pageref).
Experiments to study locomotor activity rhythms of house flies , Drosophila and syrian hamsters are proposed in the following.
Houseflies are individually kept under 12:12 h light-dark-cycles for 7 to 10 days in the containers described before. They are than recorded for 7 to 10 days in red safe light. A change between activity and rest is still found under these constant conditions. However, the period length is not 24 hours any more as was the case in the 12:12 hour light-dark-cycles, but either somewhat shorter or longer (figure 8.2) [60].
A further experiment would be the demonstration of the temperature compensation of this free run rhythm. Independent of the (constant) environmental temperature (use between 18 and 350 C) the period length is almost the same.
A number of mutants of Drosophila melanogaster are available with different period lengths. The best studied one are the per-mutants. This mutation affects eclosion rhythm and locomotor activity rhythm. per s has a shorter, per l a longer, and per 0 no rhythm. As an experiment it is proposed to compare the period lengths of these mutants with the one of the wild type. Since the Drosophila-flies are smaller then houseflies, the imaging system might pose problems. More suited is a light beam recording system which was especially developed for it. It was already described in the first part of the book. The recording cages are also different. We use plastic cuvettes as used for spectral photometers. Figure 8.6 shows the construction of a recording unit for flies. Individual flies are transferred into cuvettes containing a small piece of sugar and a wick for water supply. They are first recorded for a few days in a 12:12 hour light-dark cycle at constant temperature (e. g. 22 0 C). Afterward the recording of activity is continued under weak redlight. The activity pattern and the period length of the individual animals is determined and the mean value calculated.
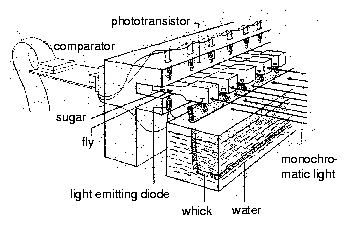
Male syrian hamster are kept individually in cages. In females the circadian rhythm is less well expressed because it is superimposed by the sexual cycle. Water is supplied from bottles with a drink nipple, hamster pellets23 serve as food.
For recording, the imaging system can be used again. If the hamster is recognized while in the vicinity of the drink nipple, the food container and in the running wheel, drinking, feeding and running wheel turning can be detected separately (see [37]).
As an experiment the dependency of the circadian rhythm from the intensity of the (weak!) continuous light can be demonstrated []. The period length and the expression of the rhythm are determined for the different light intensities (e. g. 0.1, 1, 10 lux).
We record the eclosion rhythm of Drosophila pseudoobscura flies with an imaging system. The rhythms are analysed by digital filtering. Proposals for experiments with Drosophila and mutants are made.
With the imaging system we can not only record the activity of flies, but also the eclosion rhythm of Drosophila flies. This is a population rhythm, which can be observed only in a large number of animals [137]: The eclosion of a fly out of the puparium (pupal case) happens for the individual fly only once. That this event is also controlled by a circadian clock is shown by the behaviour of the population only: Eclosion is not evenly distributed over the day, but is concentrated to special times of the day. In a 12:12 hour light-dark cycle Drosophila pseudoobscura flies eclose a few hours after onset of the light period. In a subsequent continuous dark period (or continuous red light as physiological safe light) this eclosion rhythm continues.
Drosophila pseudoobscura can be obtained from genetics departments (e.g. University of Tübingen, Germany) or from Drosophila stock centers. This species lives in dry areas in the southern part of the United States. Drosophila pseudoobscura is reared in the same way as Drosophila melanogaster (page pageref). The development takes somewhat longer (at 200 C 21 days).
The pupae are inserted individually in holes with 3 mm diameter in a rectangular metal plate using a special pair of forceps (figure 8.7). A delicate white nylon net on the lower side of the plate prevents the pupae from falling through. A sooted slide ( 50*50 mm) is put on top of the metal plate24. Allow toxic substances to dissappear from the sooted glass plate by waiting for a night.
Underneath the metal plates is a red safelight25. As soon as the flies eclose from the puparium they try to free themself. In doing so they scrape off the soot on top of the hole. They die quickly and dessicate. Redlight can now pass through the hole (figure 8.9). A video-camera is mounted on top of an air conditioned box. The lens is inserted in a hole and sees the metal plates with the pupae in the box on top of the red light. The increase of the bright spots corresponds to the course of eclosion. The number of illuminated holes (eclosed flies) is recorded (figure 8.8) hourly with the imaging system described before. The data are stored on the computer (figure 8.10). If we record 3*3 = 9 metal plates (with 10*10 = 100 holes containing pupae), we obtain for 9 groups of 100 flies each the course of eclosion in step-like curves. They can be shown as diagrams during recording on the screen.
The setup of the system is described in the first part of this book and in a handbook [2.1].
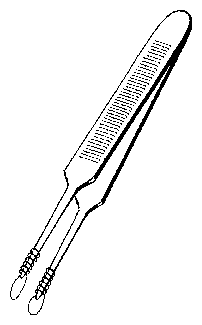
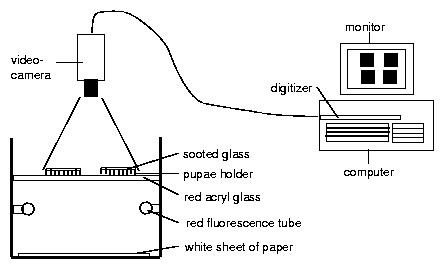

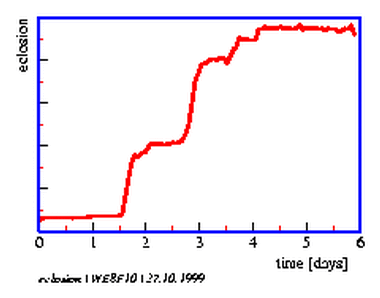
Instructions to measure the diurnal rhythm of activity and body temperature of man and to find a possible correlation with the chronobiological type (morning-, indifference-, evening type).
On an average we sleep for about 8 h per day and stay awake for 16 h. This alternation of activity and rest is caused by an oscillator which functions even in the absence of external Zeitgeber such as light-dark cycles or social contacts [6,,].
Humans staying in well-insulated isolation units or bunkers [132,] or inside caves [120] still continue to go to bed and awake on a near-daily basis thus experiencing subjective night and subjective day. Paralleling the sleep-wake cycles are the rhythms in the profiles of body temperature, urine output, concentration of ions in urine, bodily and psychological alertness [6,]. However, the period length of these rhythms, as for instance between successive maxima is not exactly 24 h any more as under normal day-night conditions. It is now typically 25 h or slightly more. This 25 h `free running rhythm' expresses itself in newborn human babies under natural day-night conditions, even though it is often punctuated by short term sleep-wakefulness cycles (figure 8.11). It is only after the lapse of the first few weeks that human babies entrain to the 24 h day-night duration of the outer world [78]. In the course of subsequent years in life humans develop tendencies to belong to 'early morning', 'late evening' or indifferent types [,].
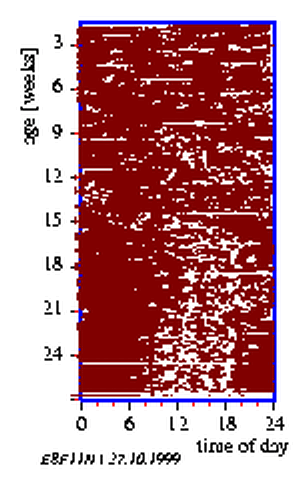
We propose to conduct a test which will show to which phase type you belong. Besides you can record your own body temperature automatically and determine temperature maxima and minima in each cycle. We could expect that the 'morning' type of persons experience temperature minima to occur earlier in the night than do 'evening' type of persons. The locomotor activity could also be measured using special recording devices.
The test is conducted in the form of a questionnaire (see page pageref). For recording body temperature we use a portable instrument with a thermoprobe which is inserted into the rectum. The rectal temperature is recorded at certain time intervals and the data stored. The wrist movements can also be recorded using the same apparatus.
The filling up of the questionnaire should pose no problems. Follow the instructions. Table 8.1 shows the evaluation of the chronobiological phase type.
| chronobiological phase type | score |
| extreme evening type | 7-10 |
| weakly expressed evening type | 11-14 |
| indifference type | 15-21 |
| weakly expressed morning type | 22-25 |
| extreme morning type | 26-31 |
A few methodological hints may be in order at this juncture. A protective covering is pulled over the thermoprobe (e. g. the disposable protective covering for fever thermometers `steritemp'). The thermoprobe consists of polyvinyl chloride with a polyvinyl cable. Therefore no organic solvents such as alcohol, aceton etc. shall be used. Medical desinfectans should not act on it too long. The probe with cable should be slowly and gently inserted into the rectum about 6cm almost until the end of the protective covering (wetten or use some cream). The best way to do this is by laying on the back and spreading the legs. The cable from the probe to the instrument should not be tight, in order to prevent the cable from braking. Relieve the cable at the plug end by using a loop. The best way to carry the instrument is on the belt of the troucers.
The function and application of the recording device are described in the instructions of the maker. The apparatus has to be equipped with a new set of batteries. The data are transferred via an interface to a PC and displayed as a function of time on screen using special software supplied by the maker.
A log note-book should be available to enter time of rising, meal times, nature and time of activities during the day, special events, time of going to bed, time of falling asleep and wakeup.
Try to find out whether there is a correlation between the time of temperature minima and the classification of the 'morning-evening' types (which is obtained from the questionnaire). This would of course mean that your own experiment and data are only one brick in many, and that a series of experiments have to be performed on numerous persons, until a clear-cut answer can be found. For this very reason it is important to maintain a detailed description of procedures and entries which is intelligible also to others and to add your results to those collected by earlier workers. This explains why the experimental strategy has been explained at greater length than those for other experiments.
After answering all questions add up the sum of individual scores. The chronobiological phase type results from the following table . Add important data such as age, sex, profession, working time, structure of day.
Use the instructions coming with the recording devices and start recording.
Record rectal temperature and activity as described for at least a week continously. It is important to note down the exact time of wakefulness, rising from bed, meals, physical activities, drinks and their nature (especially alcohol, coffee, tea, cocoa), sitting, standing, movements, lying down, going to bed etc. It is recommended to make notes elaborately, which might be needed later to interprete the recorded data and their time course.
The results can be graphically presented after the data have been entered into the computer. With a special program body temperature and activity can be displayed on the monitor screen and printed on a printer. The data can be analyzed with analysis programs.
An example for such a temperature- and activity curve is shown in figure 8.12. A rough estimation of the period length can be made from the graph.
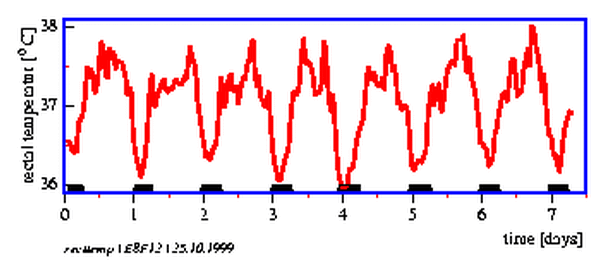
More precise is, however, the evaluation with a timeseries analysis procedure. They are described on page pageref. In order to transfer the data in this or in other programs they have to be transformed into a suitable form.
With a signal average methode an `average day' can be obtained (figure 8.13), from which the temperature minimum can be obtained. These values and the activity minimum are entered into the table 8.2 and into the graphic display (figure 8.14) below it.
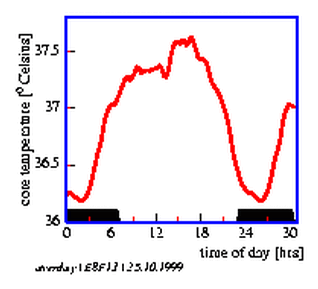
| full name | prename | M/F | age | type | temp.min. |
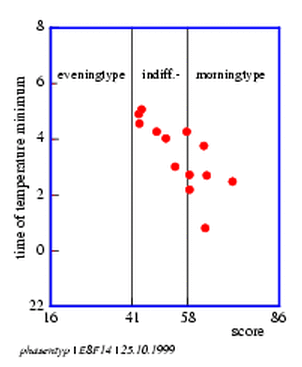
When sufficient data become available (from other persons) a correlation analysis should be undertaken. For this purpose enter the phase type score and the clock time at which the temperature minimum is found. The correlation coefficient r2 is calculated. Similarly r2 is calculated for a possible correlation between activity minima and the phase-type number. In case the data points do not permit a linear dependence of the phase-type from the time of temperature or acivity minima then other kinds of data fitting should be considered.
When data of at least 15 person are available the results should be interpreted. It is possible that there is no noticeable correlation between the chronobiological phase-types and temperature- respectively activity minima. It might be that inconsistencies occur in the data for only some persons. I.t would be of interest to look for personality traits and other factors that account for these deviations. In such instances it would be better to correlate the clear cases only and try to find simple criteria which would characterize the other `type'.
Report on the results and use also the results of other persons in order to answer the initial question (`Can chronobiological phase-types be correlated to the temperature and/or activity minima?' (page pageref) [77].
The experiment performed was supposed to answer the question whether the chronobiological phasetype, as indicated in the questionaire, was indeed related to the temperature and/or activity minima. In the case of evening phasetypes one could expect minima later in the sujective night and in the morning phasetypes earlier minima. These results are important in the context of shift work since it turns out that markedly morning phasetype persons are unsuited for night shift work [1,,] .
There might emerge other practical applications to these findings. There are indications that the chronobiological phasetypes may in some cases arise as a function of age and that fluctuations are found during the age children go to school . Unfortunately our society does hardly take any notice of these phenomena. In fact many traffic accidents involving school children might be avoided by adjusting school timing to suit the chronobiological behaviour traits of children. Obviously the alertness and performance skills of children are also very strongly correlated to the chronobiological phase types. This can be demonstrated easily by experiments. A vast array of experimental possibilities lies in this area of chronobiology. Someone remarked that more marriages might break up because of chronbiological phase incompatibilities of the partners than of other reasons. Watch out!
Besides body temperature and bodily activity several other physiological processes display a distinct circadian rhythmicity in humans. Those who wish to investigate a few of these are referred to Koukkari et al. [82]. Even body temperature and activity can be used to answer a series of other questions, as for example:
This list consists
of questions which relate to your activity and feeling of being awake in the
morning and evening. In answering questions 1 to 4 you should assume that
you have to work during the day for 8 hours at a selfselected time. Answer all
questions honestly. Place only one cross for each question.
How difficult would it be for you if you had to go to bed all times
at 01:00 o'clock?
How difficult would it be for you if you had to rise up in the morning all times at 06:00 o'clock?
Imagine you decided to participate in a fitness training. Your friend proposes an hours training twice a week. For her/him the best time in the morning would be from 7 to 8 o'clock. How would this be with you?
Imagine you decided to participate in a fitness training. Your friend proposes an hours training twice a week. For her/him the best time in the evening from 23 to 24 o'clock. How would this be with you?
Underline at which time you normally go to
bed.
Example:
¯20:00 ¯21:00 ¯22:00 ¯23:00¯24:00 ¯01:00 ¯02:00
¯03:00
20:00 21:00 22:00 23:00 24:00 01:00 02:00 03:00
Underline at which time you normally rise up
from bed.
¯05:00 ¯06:00 ¯07:00 ¯08:00 ¯09:00 ¯10:00 ¯11:00 ¯ 12:00
Are you a morning- or evening active person?
The evaluation of the questionaire is on page (siehe Seite pageref)
Overview:
Quite a number of organisms use daylength for photoperiodic reactions.
In this way they are able to adapt themself to changes in environmental conditions
during the course of a year. Experiments for photoperiodic reactions in Drosophila
flies and for flower induction in short day plants are described.
Organisms living in temperate or higher latitudes of the earth had to adapt themself to the seasonal changes of the environment. They mostly use the daylength as the most reliable indicator of the time of year. If in the fall the days become shorter, it signals that winter is coming and with it unfavorable life conditions. The organisms have than still time to prepare for it, by inducing a special behaviour (e. g. larvae crawl into the soil) or metabolic processes (for instance in insects protection of the body fluid against freezing).
There are numerous examples for photoperiodic reactions. In plants the daylength often determines whether flowers are formed. In plants of temperate or higher zones this occurs usually in long day (e. g. shooting of salad). But there are also short day plants found. In their case flowering is induced by short day. To them belongs the Crassulacea Kalanchoe blossfeldiana from arid areas of Madagaskar.
Many insects survive unfavorable conditions in the state of diapause. Diapause is also in most cases photoperiodically induced.
In mammals the colour of the fur can be controlled photoperiodically. The sibirian hamster is white in the winter and brown in the summer. Another example: The rutting season is induced by certain daylengths. For this and further examples see [8].
Photoperiodic reactions have an important practical significance in agriculture (chicken for instance lay eggs only in long day. By using additional light in the night egg is maintained also in the winter). Furthermore, in horticulture photoperiodic treatment is used to induce flower induction at certain times of the year when those plants would usually not flower.
Quite a number of plants can be induced to flower by short days. The morning glory Pharbitis nil is especially useful for demonstration, because in the strain `violett' a single short day is sufficient to induce the photoperiodic reaction. Furthermore this plant can be induced already in the seedling stage. It takes only a few days until the photoperiodic reaction is induced. This allows to conduct experiments in a short time.
Seeds can be purchased from the Maruthane Trading Company in Tokyo (Japan). They can be kept in the freezer for a few years. The seeds are treated with concentrated sulfuric acid (45 Minuten), the acid poored off (in much water, attention, never poor water in the concentrated acid, danger of splashing!) and much tapwater added to the seeds. They are heated for a short time which synchronizes germination. After 12 hours of watering in running tap water (with a net on top of the seeds to prevent the seeds from floating away) plant the seeds 15 mm deep in garden soil in pots and allow to germinate in continuous light.
After the cotyledons have unfolded themself the plants can be induced photoperiodically. A single darkperiod is sufficient in the strain `violett'. Afterward the plants are transferred again in continuous light. Vary the length of the dark period for groups of 10 plants between 6 and 20 hours.
Already one week after the dark treatment the condition of the buds can be determined under the binocular. Flower buds have two long bracts und a broad apex, whereas vegetative buds possess a pointed apex (figure 9.1).
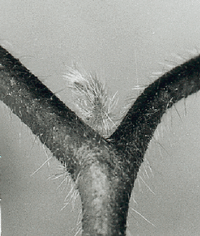

Determine the mean number of flowers per plant in each group. Plot the results against the length of the dark period. The critical dark period is the length where the plants show 50% of the maximally inducible flowers (figure 9.2).
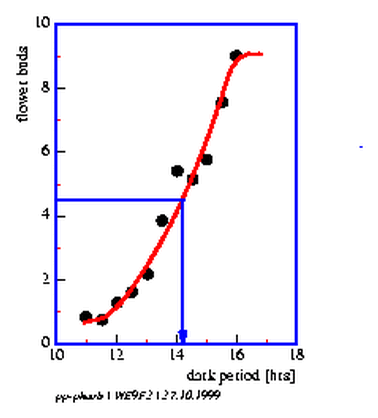
The experiments can be performed at another environmental temperature in order to test, whether the critical dark period is influenced by it. Remember that circadian rhythms are temperature compensated in their period length. According to Bünning[] the daylength measurement in photoperiodic reactions occurs with the help of the circadian clock. Accordingly the critical daylength should be independent of the environmental temperature.
In numerous insects developmental steps are photoperiodically controlled. Take the development of ovaries of female Drosophila litoralis as an example. It is blocked in short day. Cultures are kept at the department of genetics, SF 90570 Oula (Finland). There are various strains, the critical daylengths of which differ. Strain 1008 from the Tessin has no diapause (`Ticino'), strain 1036 has a critical daylength of 20 hours (`Oulu 1A'), im-3 (`Kutaissi'), and 1052 (`Batumi') of 12 hours. The further south the species is found, the shorter is the daylength at which ovary development is stopped.
Rear in 250 ml bottles. For oviposition to occur flies must be at least 2 weeks old. Wash the larvae out with water, bring them in small quantities in vials which have to be kept humid and supplied with yeast. Use cotton- or cellulose plugs. Food recipe see [85]. Nipagin prevents mold growth. At 19 to 20 0 C the development from oviposition to eclosion takes 3 weeks. Eclosion is spread over a week. Change food every fourth day, 100 flies per vial. Transfer flies with exhaustor (figure 9.3).
The adults are photoperiodically sensitive [85,]. After eclosion every second day flies are collected and transferred into different photoperiods at 16 0 C. For instance 6, 9, 12, 13.5, 15, 16.5, 18, 21 h light per day. The best method is to use light tied boxes with lids in continuous light. They are closed for the dark periods. The opening and closing of the lids can also be automatically controlled by a motor via an electric timer.
After (2-) 3 weeks the females are checked for the development of the eggs (the males are easily recognizable by their red testes). Pull with two pointed forceps the abdomen apart and check at a magnification of 25* the egg dotter under a drop of water. The developed ovaries are much larger. It takes about 15 minutes to check 100 flies.
For each record about 200 flies from each culture are needed. Enter the results in a table and plot the values graphically (% diapause as a function of length of light period).
This part of the book is somewhat preliminary and will be worked at in the future. I consider it important, since the development of the field of biological rhythms depends on the young people coming in contact with it, and on the interest of the public.
In the first chapter of this part, books are listed and commented on which introduce the interested layman into the field. Proposals are made for projects in adult education courses and teaching material is mentioned.
In the next chapter projects for schools are described on two levels. The lower level is aiming at pupils and students who want to be introduced into the field and to back it up with observations, simple experiments and modelling. At the advanced level more elaborate projects are introduced and weight is put on own planning and execution of experiments. In this chapter reference is frequently made to methods described in part I and examples given in part II.
A third chapter is concerned with teaching this field at the university level, where I have much own experience. Lecture topics, seminar proposals and -styles, are briefly mentioned.
In the last chapter didactic considerations will be discussed.
Furthermore, instruments, recording systems, laboratory material, sources for material and organisms, handling and rearing of unicells, plants and animals which have been proposed to be used in this book are described.
Overview:
There are a number of books which introduce into the area of chronobiology.
Some of them are listed in the following section and their content is summarized.
In a further section topics for small projects are proposed which interested
laymen could tackle for instance in the frame of an adult education course.
The chapter closes with hints for audio- and visual material.
The following books are useful as an introduction to chronobiology and their content is summarized briefly:
The following topics are useful for smaller projects :
Flowers might open and close in a diurnal manner. The movement can be brought about by turgor changes in the cells of the petal. Examples are the Kalanchoe petal movements we have heard about already (page pageref). The petals of water lillies do also move in a circadian way. Even cut stripes of the petals show this movement [].
Some flowers open at certain times of the day. This is especially impressive in Oenothera. The process of opening occurs in a few minutes. An interesting project is to determine the time of opening as a function of time of the year (and consequently as a function of daylength). The first flowers open in the begin of july, the last one in october or even november. Is the time of day at which they open influenced by temperature, clouds, wind? The flowers of morning glory Pharbitis open also at certain times.
The opening of flowers is often connected with fragrance . Other flowers such as Exaccum are open all the time, but the fragrance occurs rhythmically. One should also test whether perhaps the sensitivity of the human nose and not the fragrance of the flowers changes rhythmically. For this purpose fragrant substances can be smelled in different concentrations at different times of the day. The threshold value (lowest still recognizable concentration) should be constant, if the sensitivity of the nose does not change. Many petal movements or openings of flowers or fragrance productions of plants are connected with the attraction of insects, to ensure fertilization. This offers also the opportunity to identify the fertilizing insects.
The algal suspension in the test tube is observed every 3-4 hours and the density recorded. In many algae a substantial part of the population settles during the night at the bottom and during the day they are more or less uniformly distributed in the medium. The cause for it is in the case of Euglena a difference in beat activity of the flagellae respectively a loss of flagellae during the night. There are, however, also indications of diurnal vertical migrations based on a different density of the individual cell [46]. These events can be documented by photography or video pictures. In this way it is easier to compare the density differences in the various parts of the test tube.
Especially interesting would be studies on bluegreen algae (Cyanobacteria). It was only in the last years that diurnal and circadian rhythms were described in these prokaryotes [66]. These rhythms show up in photosynthesis, nitrogen fixation and carbohydrate . Other movements in Cyanobacteria are perhaps also controlled in a circadian manner. Cyanobacteria show vertical movements due to density differences. To determine Cyanobacteria see [15].

Adresses of groups working in the field of chronobiology for Germany can be found in the internet under Roenneberg, University of M�nchen, Institute for physiological Psychology. In the USA is the Center of Biological Timing in Charlottesville, University of South Carolina. University calendars or Internet pages on research fields in universities are usefull. Publications of the groups help also to become familiar with the work done there.
Overview:
Some books are mentioned which in addition to the one of the last chapter
introduce into the area of chronobiology. Ten chronobiology topics for an advanced
course at schools are proposed.
As an introduction the following books and articles are suited:
A multimedia show will be composed which aims at motivating the student for this field. It will offer an overview of the different rhythms at different organisms from unicellulars to man.
Topics from the field of chronobiology are well suited for an advanced course at schools. In the following, ten units are described with content, time necessary to do them, and the kind of instruments needed.
Slides, movies, videos and suitable computer programs are mentioned in the last chapter of this part (`teaching aids').
Overview:
Lectures, seminars, courses as introductions into chronobiology at universities,
research projects, research groups.
The field of chronobiology can be offered in courses, seminars, colloquia and lectures. Details are found in the catalogue of lectures of the universities. See also page pageref.
I have experience of many years in teaching chronobiological topics. In a lecture cycle `adaptation of organisms to the time structure of the environment' I have covered in five terms the following topics:
The content of these lectures will be presented on the Internet (see page 10.1). Here are some short overviews of the content:
One of the main obstacles in preparing these lectures was the lack of more recent books and review articles. Therefore one had to use quite often original papers, to select the most important findings and to try to cover the newest results of this rapidly expanding field.
Seminars are useful to introduce into the field of chronobiology. I have used different strategies and found the Epstein-method [44] useful. All the participants read the same publication at home and it is discussed in the seminar. The basic idea is, that an original paper reflects quite accurately the way of scientific working of a scientist. This strategy stresses mainly the methods used and the way of presenting the results, whereas the subject content of the publication is less important. In this way the learned is transferable to other fields in biology and natural sciences.
I have also offered seminars in which teaching aids were produced by the participants, and some of them are referred to in this part. This kind of seminar is especially interesting for students which want to become teachers.
Courses are the most effective teaching arrangements to get to know the field of chronobiology. At the university of Tübingen we propose projects which can be worked at in small groups. The students have 2 to 3 weeks time for the practical studies. They are, however, already familiar with the field and the methods by an introductory seminar. It is of much value to work on projects which have not been studied so far. The knowledge to work at the frontier of science balances the danger, that such a study might also lead to an unexciting result. It is, however, necessary to have the recording methods and other aids available. Otherwise too much time has to be invested which is then lacking for the studies themself. One reason to write this book was to offer the necessary prerequisites for this.
Often research studies in the field of chronobiology are started as a consequence of working with such a project in a course. A number of students become so motivated that they decide to continue the project in form of a diploma-, teachers examination- or even doctoral thesis.
How to find a topic or project has been described already before (page pageref). A list of groups working in the field of chronobiology is found on the Internet (page pageref). Lecture catalogues or lists of research activities at universities are of help as is the Internet. Using the publications of the research groups allow to get an overview of the work done in these groups.
Overview:
Learning objectivs are briefly mentioned. Afterward we will list aids
for the teaching of chronobiology such as programs, movies, slides, instruments,
laboratory material, supply sources and rearing of experimental organisms.
Didactic aspects are important in schools, adult education courses and universities. This applies for the field of chronobiology to the same extent as for other fields of natural sciences. I will mention here just briefly some high level teaching aims, which are essential, and refer to literature [118,].
Important teaching aims:
To reach these goals, a number of aids have to be offered such as
To offer learning- and working places and to provide resources is, especially for research work, the most elaborate task.
As teaching aids the following should be available:
The aids described in the book and the supply sources are compiled in the following:
Movies from the catalogue for scientific movies of the `Institut für den wissenschaftlichen Film' (IWF) (Göttingen) (partly available in english versions also).
Remark: in italics latin names and references to other entrances in the glossary
|
1 Thanks to D. Engelmann (help with computer work), J. Dittami and P. Reinhard (proofreading), Schneider-Uhle (figures) and students (feedback). This book was typeset by LYX (http://www.lyx.org), a powerful document processor using the LATEX typesetting system.
2All our education is not worth a penny if courage and joy are lost.
3how oscillations are characterized is described on page pageref and illustrated in figure 3.1
4if you do not understand terms, check in the glossary at the end of the book
5words teach, examples bring you forward
6sometimes available in drug stores and gas stations
7The Q 10 value is a measure of the temperature dependency of a process
8if necessary cut out a page of the book to compensate for thickness
9Medline (http://www.biomednet.com), Biological Abstracts, Swets and Zeitlinger with the contents of more then 14000 journals (http://www.swetsnet.nl/direct)
10address: Buchenweg 27, D72820 Sonnenbühl(Germany)
11For this purpose the oscillation is normalized to an evident measure, e.g. to 3600 or 2p circumference of the unit circle or to 24 hours circadiane time. 1800 , p or 12 CT as phase references would all mean, that half of the oscillation has passed.
12there are further procedures for smoothing, in which the values of a smoothing window are weighted; the center value could for instance obtain a high weight, the neighbouring values a smaller weight, and the peripheral values a small weight. Digital filters weight with functions (page pageref)
13supplied by the `CoMet Verlag für Unterrichtssoftware, Duisburg'
14Dr. Diez-Noguera, Group de Cronobiologia, Laboratori de Fisiologia, Facultad de Farmacia, Av. Diagonal 643, SP 08028 BARCELONA (SPANIEN)
15available from the Marutane Trading Co. in Kyoto, Japan
16green fluorescence tubes Philips TL40W/17 covered with green foil nr. 39 Cinemoid
17e.g. plasticine
18If the offset of the recorder is not sufficient, use counter-voltage, to reduce the signal from the amplifier. This allows to switch to a smaller voltage range
19Fa. Stereo Optik, Mainstr. 13, D63128 DIETZENBACH, Tel. 06074 27222
20see study 4.
21substitute of agar from indian plantain-seed pots
22red fluorescence tube with red Cinemoid foil nr. 6, primary red
23e. g. Altromin company
24soot with a candle. Cheap candles make more soot and are in this case to be preferred.
25Philips TL20W/15 with three layers of red Cinemoid foil nr. 6 and one layer of yellow foil nr. 5a.